Raising the Bar for Hemophilia Management
- 1. Raising the Bar for Hemophilia Management Guy Young, MD Director, Hemostasis and Thrombosis Center Children’s Hospital Los Angeles Professor of Pediatrics University of Southern California Keck School of Medicine Robert F. Sidonio, Jr., MD, MSc Professor of Pediatrics Medical Director Hemophilia of Georgia Center for Bleeding and Clotting Disorders Children’s Healthcare of Atlanta
- 2. Provided by i3 Health ACCREDITATION In support of improving patient care, i3 Health is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team. i3 Health designates this live activity for a maximum of 1.25 AMA PRA Category 1 Credits . Physicians should claim only the credit commensurate with the extent of their participation in the activity. A maximum of 1.25 ANCC contact hours may be earned by learners who successfully complete this nursing continuing professional development activity. i3 Health has approved this knowledge-based activity for 1.25 ACPE CE hours. The Universal Activity Number for this activity is JA0008128-0000-25-024-L01-P. Pharmacy CE credits can be submitted to the NABP upon successful completion of the activity and the evaluation by providing your NABP ID and DOB, which must be submitted within 60 days of completion. i3 Health has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credit for activities planned in accordance with AAPA CME Criteria. This activity is designated for 1.25 AAPA Category 1 CME credits. Approval is valid until 8/23/2025. PAs should only claim credit commensurate with the extent of their participation. INSTRUCTIONS TO RECEIVE CREDIT An activity evaluation link or paper evaluation will be available at the conclusion of this activity. To claim credit, you must submit a completed evaluation at the conclusion of the program. Your certificate of attendance will be available for download after submission or emailed to you within 7 days. UNAPPROVED USE DISCLOSURE This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The planners of this activity do not recommend the use of any agent outside of the labeled indications. The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of the planners. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings. DISCLAIMER The information provided at this CME/NCPD/CPE activity is for continuing education purposes only and is not meant to substitute for the independent medical/clinical judgment of a healthcare provider relative to diagnostic and treatment options of a specific patient’s medical condition. Unless otherwise noted, i3 Health, the host of this industry-sponsored educational symposium/networking event, is solely responsible for its content, including any information presented or distributed during the event. The National Bleeding Disorders Foundation does not endorse any particular treatment products or manufacturers; any reference to a product name is not an endorsement by NBDF. NBDF is not a regulatory agency and cannot make recommendations relating to the safety of manufacturing of specific treatment products. For recommendations of a particular product, the regulatory authority in a particular country must make these judgments based on domestic legislation and national health policies. COMMERCIAL SUPPORT This activity is supported by an independent medical education grant from Sanofi.
- 3. Disclosures Guy Young, MD Consultant: Alnylam, ASC Therapeutics, Bayer, BioMarin, CSL Behring, Genentech/Roche, HEMA Biologics, Novo Nordisk, Octapharma, Pfizer, Sanofi, Spark, Takeda Grants/research support: HEMA Biologics, Sanofi Robert F. Sidonio, Jr., MD, MSc Advisory board or panel: Bayer, HEMA Biologics/LFB, Hemab, Novo Nordisk, Octapharma, Pfizer, Sanofi/Sobi, Star, Takeda Consultant: Bayer, HEMA Biologics/LFB, Hemab, Novo Nordisk, Octapharma, Pfizer, Sanofi/Sobi, Star, Takeda Grants/research support: HEMA Biologics/LFB, Octapharma, Takeda i3 Health has mitigated all relevant financial relationships
- 4. Learning Objectives Define appropriate treatment goals and targets for the management of hemophilia A and B Evaluate the clinical utility of novel and emerging prophylactic agents for the treatment of individual patients with hemophilia A and B Assess methods to improve the early detection of hemophilic arthropathy development and progression Apply strategies to enable increased physical activity for patients with hemophilia while minimizing risk of bleeds
- 5. Patient Case: Hemophilia A 14-year-old male with severe hemophilia A with history of transient inhibitor Baseline FVIII <1% Initially started on SHL FVIII Trial of emicizumab for 12 months; developed a large muscle bleed but wants to know his options VIII = factor VIII; SHL = standard half-life.
- 6. Polling Question What is known about muscle bleeds in patients on emicizumab? a. Emicizumab does not prevent muscle bleeds b. Emicizumab is only effective against joint bleeds c. There are reports of serious muscle bleeds especially in teenagers and young adults on emicizumab d. Factor concentrates are proven to be more effective against muscle bleeds than emicizumab
- 7. Patient Case: Hemophilia B FIX = factor VIX; rFIX:Fc = recombinant factor IX Fc fusion protein; ABR = annualized bleeding rate. 19-year-old male with severe hemophilia B without history of inhibitor Baseline FIX <1% Initially started on SHL FIX product 3 times a week Recently attempted rFIX:Fc weekly with good trough at Day 7 (2%) and ABR ~3 Active in soccer (promises doesn’t head the soccer ball) Wants to consider alternative therapy as he is getting “tired” of frequent injections
- 8. Polling Question TFPI = tissue factor pathway inhibitor; EHL = extended half-life; siRNA = small interfering ribonucleic acid. What are the novel options for prophylaxis for a young adult with severe hemophilia B? a. Factor replacement (SHL and EHL products) are the only option b. An anti-TFPI subcutaneous drug such as concizumab or marstacimab c. An siRNA subcutaneous option such as fitusiran monthly or every 2 months d. Options b and c are reasonable for prophylaxis
- 9. Hemophilia A and B: Scope of the Problem and Unmet Needs
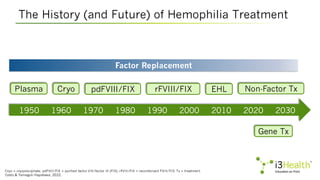
- 10. 1950 1960 1970 1980 1990 2000 2010 2020 2030 Factor Replacement Plasma Cryo pdFVIII/FIX rFVIII/FIX EHL Non-Factor Tx Gene Tx The History (and Future) of Hemophilia Treatment Cryo = cryoprecipitate; pdFVIII/FIX = purified factor VIII/factor IX (FIX); rFVIII/FIX = recombinant FVIII/FIX; Tx = treatment. Ozelo & Yamaguti-Hayakawa, 2022.
- 11. Why Do We Need New Therapies for Hemophilia?
- 12. Hemarthrosis Images courtesy of Guy Young, MD. The most common long-lasting sequelae of hemophilia is permanent joint disease called hemophilic arthropathy Acute hemarthrosis
- 13. Synovitis Images courtesy of Guy Young, MD.
- 14. Arthropathy Images courtesy of Guy Young, MD. How effective is prophylaxis at preventing hemophilic arthropathy? End-stage arthropathy
- 16. Longitudinal Comparison: Early and Delayed Prophylaxis CPJAS = Colorado Pediatric Joint Assessment Scale; JOS = joint outcomes study; JOS-C = joint outcome study continuation. Figure adapted from Boulden Warren et al, 2020.
- 17. ~33% up to 60% Joint Bleeds Still Common, Even Among Patients on Prophylaxis Wilkins et al, 2022; Srivastava et al, 2020. of adult patients experienced breakthrough joint bleeds despite prophylaxis treatment of pediatric patients experienced breakthrough joint bleeds despite prophylaxis treatment Breakthrough joint bleeds can have detrimental effects on joint health In a United Kingdom observational study over 1 year:
- 18. Joint Bleeds Can Cause Damage Via Multiple Pathways Kizilocak & Young, 2019; Calcaterra et al, 2020. A vicious circle of bleeding can occur—leading to permanent joint damage 1 Iron released into synovial fluid causes chemical damage via inflammatory cytokines and chemokines 2 Synovium inflammatory damage and degenerative cartilage changes occur 3 New friable vessels, which are more prone to bleeding, form via neovascularization Bleeding Synovial inflammatory damage/ degenerative cartilage changes New friable vessels via neovascularization Iron accumulation
- 19. FVIII Prophylaxis Delays Hemophilic Arthropathy Manco-Johnson et al, 2007; Srivastava et al, 2020. People with hemophilia A who receive FVIII prophylaxis show fewer imaging markers of joint damage Patients with severe FVIII deficiency initiated on early FVIII prophylaxis have better long- term outcomes than those who started later Initiation of prophylaxis is recommended in early childhood, ideally before 3 years of age Markers of joint health in a cohort of children receiving FVIII prophylaxis or on-demand treatment 0 10 20 30 40 50 0 5 10 15 20 2/27 13/29 1/28 5/27 P=0.002 Patients with joint damage (%) Patients with joint damage (%) MRI Radiography P=0.10 Prophylaxis On- demand Prophylaxis On- demand
- 20. Regular FVIII Infusions Protect Bone and Joint Health HJHS = Hemophilia Join Health Score. Gamal Andrawes et al, 2020. People with hemophilia A who receive FVIII prophylaxis show better HJHS and bone status FVIII prophylaxis is associated with improved bone and joint status compared with on- demand treatment In hemophilia A, clinical assessment of joint structure and function is commonly performed using the HJHS in children and adolescents 0 20 40 60 80 100 0 1 2 3 4 5 6 7 8 P=0.022 P=0.035 23/35 5/15 HJHS and bone status in children with severe hemophilia A who received low-dose FVIII prophylaxis (n=35) or on-demand (n=15) treatment Median HJHS Patients (%) Patients without bone deformity HJHS Prophylaxis On- demand Prophylaxis On- demand
- 21. Musculoskeletal Ultrasound (MSKUS) Foppen et al, 2018; Bakeer & Shapiro, 2019. Can Be Used to Assess Joint Health and Identify Source of Pain MSKUS can be used to determine whether acute musculoskeletal pain is due to arthritis or is bleed-related MSKUS is an accurate and available alternative to MRI Benefits vs conventional MRI: • Ultrasound can accurately assess synovial hypertrophy, bone surface irregularities and cartilage abnormalities in hemophilia patients with limited joint disease • Can be used for routine evaluation of early joint changes • For hemarthrosis evaluation, MSKUS detected low concentrations of intra-articular blood (as low as 5%) and discriminated between bloody and non-bloody fluid
- 22. Additional Methods to Assess Joint Bleeds and Joint Health Gouw et al, 2019; St-Louis et al, 2022. Joint Health Measurement Age Physical Examination Scores World Federation of Hemophilia Orthopaedic Joint Score (WFH) Children and adults • Hemophilia-specific physical examination for joint health of the knees, ankles, and elbows • An additive score ranging from 0-12 for knees and ankles and 0-10 for elbows, with 0 being an unaffected joint and 10 or 12 being most affected • The maximum total score is 68 without the pain score and 86 with the pain score (0-3 for each joint) • Used to develop the CPE, PJS, and HJHS Colorado Physical Examination Scale (CPE) Children • The original CPE assessed the knee, ankle, and elbow to detect subtle abnormalities in joint health • Scores range from 0-31 for ankles/knees and 0-29 for elbows • Higher scores indicate worse joint health The Petrini Joint Score (PJS) Adapted from the WFH scoring system for use in children • 3 points can be added when the joint is considered a target joint and when chronic synovitis is present • Ankles, knees, and elbows are assessed • Scores range from 0-25 per joint, with a maximum total score of 150 • Higher scores indicate worse joint health Hemophilia Joint Health Score (HJHS) Originally designed for children, but now validated in adults • Originally designed to detect mild joint impairment in knees, ankles, and elbows • Current version (2.1) assesses swelling, duration of swelling, muscle atrophy, crepitus on motion, flexion loss, extension loss, joint pain, global gait, and strength of the knees, elbows, and ankles • Incorporates scoring of the WFH, CPE, and PJS • The maximum score is 124, with a higher score indicating worse joint health (version 2.1) • Version 1: max score was 148 Despite existing gaps in the evidence for all measurements, HJHS properties were most extensively studied and are used in clinical practice in young adults treated intensively and in children
- 23. Why Do We Need Nonfactor Therapies?
- 24. Prophylaxis Schedule: Month Slide courtesy of Guy Young, MD. Sunday Monday Tuesday Wednesday Thursday Friday Saturday
- 25. Prophylaxis Schedule: Year Slide courtesy of Guy Young, MD. And that is just the first year…
- 26. IV = intravenous. Prophylactic Schedule With Extended Half-Life Factor VIII Sunday Monday Tuesday Wednesday Thursday Friday Saturday 104 IV injections per year
- 27. Parents Don’t Like It…Kids Hate It! Image courtesy of Guy Young, MD. Prophylaxis for Hemophilia And this is a good day, Dr. Young
- 28. This was for 1 infusion!! Prophylaxis for Hemophilia Image courtesy of Guy Young, MD.
- 29. Callaghan et al, 2018. Improve joint outcomes By reducing bleeding events including subclinical bleeding Reduce the treatment burden For intravenous therapies, reduce the frequency of infusions Subcutaneous administration (most new medications and those being developed are given subcutaneously) Oral administration—today this is still just a dream Eliminate the treatment burden—gene therapy? Improve quality of life Allow for patients to live a “hemophilia-free life” Goal of New Therapies
- 30. AAV = adeno-associated virus. Callaghan et al, 2018; Miesbach et al, 2022. Novel factor therapies Efanesoctocog alfa Nonfactor therapies FVIII mimetics Rebalancing agents Gene therapy AAV transfer Lentiviral transfer Gene editing Categories of Novel Therapies
- 31. XI XIa Ca++ IX IXa Ca++ X Xa Ca+ + VIIIa VIII VII VIIa TF Xa X Ca++ Prothrombin (II) Thrombin (IIa) Fibrinogen (I) Fibrin XIII XIIIa Cross-linked fibrin V Va Thrombin (IIa) Antithrombin Protein C Protein Ca Thrombin (IIa) Protein S Protein S Thrombin (IIa) Ca++ Tissue factor pathway inhibitor (TFPI) Emicizumab MIM8 NXT007 Concizumab Marstacimab MG1113 Fitusiran Serpin PC Anti-APC Mab Anti-PS Mab Anti-PS siRNA Anti-PS Mab Anti-PS siRNA Efanesoctocog alfa Gene therapy Gene therapy PS = phosphorothiotate; Mab = monoclonal antibody; TF = tissue factor; Ca = calcium; APC = antigen-presenting cell. Young, 2023. Novel Therapeutics Mechanisms of Action Inhibits Replacement therapy Substitution therapy Rebalancing agents
- 32. Improving on Current Factor Therapy
- 33. Novel Factor Therapy Lissitchkov et al, 2022. MOA Drug Dosing Regimen Development Phase Comments Factor replacement (non-inhibitor patients) Efanesoctocog alfa (efa) IV weekly Approved Higher factor levels throughout the dosing interval (~70%-15%) than current factor products
- 35. Structure of BIVV001 VWF = von Willebrand factor. Konkle et al, 2020.
- 36. BIVV001 Efficacy: Factor IVVV Activity at 14 Days Konkle et al, 2020. Group Receiving Dose of 65 IU/kg
- 38. 0 0.5 1 1.5 2 2.5 3 3.5 Annual Bleed Rate SOC Prophylaxis Efanesoctocog alfa Primary Endpoint Efanesoctocog Alfa Clinical Results SOC = standard of care. von Drygalski et al, 2023. SOC Prophylaxis Efanesoctocog alfa
- 39. von Drygalski et al, 2023. No serious adverse effects No inhibitors No thrombosis No allergic reactions Efanesoctocog Alfa Safety
- 40. Nonfactor Therapies Young, 2023. They are all given subcutaneously and most of them less/much less frequently than factor therapy They are (based on trial data) more effective at preventing bleeding than factor therapy They therefore may be more effective at preventing joint disease
- 41. Slide courtesy of Guy Young, MD. XI XIa Ca++ IX IXa Ca++ X Xa Ca+ + VIIIa VIII VII VIIa TF Xa X Ca++ Prothrombin (II) Thrombin (IIa) Fibrinogen (I) Fibrin XIII XIIIa Cross-linked fibrin V Va Thrombin (IIa) Antithrombin Protein C Protein Ca Thrombin (IIa) Protein S Protein S Thrombin (IIa) Ca++ Tissue factor pathway inhibitor (TFPI) Emicizumab MIM8 NXT007 Concizumab Marstacimab MG1113 Fitusiran Serpin PC Anti-APC Mab Anti-PS Mab Anti-PS siRNA Anti-PS Mab Anti-PS siRNA Efanesoctocog alfa Novel Therapeutics Mechanisms of Action Inhibits Replacement therapy Substitution therapy Rebalancing agents
- 42. Factor VIII Mimetics (For Hemophilia A Only) SC = subcutaneous; Q = every; prophy = prophylaxis; Hem = hemophilia. Hemlibra® prescribing information, 2025; Hermans et al, 2024; Shima et al, 2025. MOA Drug Dosing Regimen Development Phase Comments Substitute for the function of activated FVIII Emicizumab SC Q1, 2, or 4 weeks Approved Most commonly prescribed medication for prophy in Hem A Mim8 SC Q1 week or Q1 month 3 Preclinical studies show increased thrombin generation compared to emicizumab NXT007 SC Q1, 2 or 4 weeks? 1
- 43. What Do We Mean by Mimetic?
- 44. Factor VIII Mimetics (Hemophilia A Only) Kitazawa et al, 2012.
- 45. Emicizumab: Efficacious in Severe + Non-Severe Hemophilia A Callaghan et al, 2021. • With nearly 3 years of follow-up, low bleed rates were maintained with emicizumab prophylaxis • After Week 24, at least 97% of participants had ≤3 bleeds in each treatment interval • Emicizumab remained well-tolerated over long- term follow-up
- 47. Severe Muscle Bleeds in Hemophilia A Patients on Emicizumab? BE = bleeding event. Batsuli et al, 2023. Median (range) age at time of BE Inhibitor patients: 13 (13-27) years Non-inhibitor patients: 14 (10-21) years Iliopsoas (n=3) Calf (n=3) Sites of muscle bleeds Non-inhibitor patients (n=11) Inhibitor patients (n=4) Thigh (n=6) Forearm (n=1) Lower leg (n=1) Iliopsoas (n=2) Thigh (n=2) Forearm (n=1) BEs (n=14) BEs (n=5) Thigh and iliopsoas were the most common sites of the muscle bleeds 7 2 Spontaneous 6 Sports/activity– related Weight-lifting– related 4 Other trauma- induced Type of bleed 19 severe muscle bleeds occurred in the 15 hemophilia A patients
- 48. Mim8 HA = hemophilia A; CFC = clotting factor concentrates. Lillicrap et al, 2024; Mancuso et al, 2024; Østergaard et al, 2021; GlobeNewswire, 2025. IgG anti-FIXa/anti-FX bispecific antibody with unique binding sites 13-18–fold enhanced activity compared to emicizumab FVIII bioequivalence up to 36.8% (assay-dependent) FRONTIER2 Phase 3, adolescents and adults with HA with or without inhibitors Improved ABR with either weekly or monthly dosing compared to prior CFC prophylaxis FRONTIER3 interim analysis with 74% of children with 0 treated bleeds on weekly injections
- 50. Activity of Mim8: Peak Thrombin Generation biAb = bispecific antibody; SIA = sequence identical analogue. Østergaard et al, 2021.
- 51. FRONTIER2: Mim8 Efficacy QW = every week; QM = every month. Mancuso et al, 2024. FRONTIER2: phase 3 trial of Mim8 in 254 patients ≥12 years of age with hemophilia A with or without inhibitors Mim8: investigational FVIII mimetic that is approximately 15 times more potent in vitro than emicizumab
- 52. FRONTIER2: Mim8 Efficacy (cont.) ad/c in Arm 2a due to nerve paralysis of probably relation to Mim8; d/c in Arm 3 due to sleep disorder and dyspepsia possibly related to Mim8 and coronary artery disease unlikely to be related to Mim8. Mancuso et al, 2024. Safety Summary No evidence of neutralizing antibodies targeting Mim8 Rates of injection-site reactions were low (9.5% in Arm 2a, 5% in Arm 2b, 12.2% in Arm 3, and 8.2% in Arm 4) Pre-Study On-Demand Treatment Group Pre-Study CFC Prophylaxis Group Arm 1, On Demand (n=17) Arm 2a, Mim8 QW (n=21) Arm 2b, Mim8 QM (n=20) Arm 3, Mim8 QW Arm 4, Mim8 QM (n=98) Run-in (n=98) Main (n=98) Run-in (n=98) Main (n=98) Total exposure time, yr 8.41 9.94 9.93 77.07 46.62 73.76 48.7 All AEs, n (%) 5 (29.4%) 10 (47.6%) 6 (30.0%) 58 (59.2%) 71 (72.4%) 51 (52.0%) 65 (66.3%) Serious AEs, n (%) 1 (5.9%) 1 (4.8%) 0 6 (6.1%) 4 (4.1%) 2 (2.0%) 4 (4.1%) Thromboembolic events, n (%) 0 0 0 0 0 0 0 Severe AEs, n (%) 1 (5.9%) 2 (9.5%) 0 1 (1.0%) 2 (2.0%) 2 (2.0%) 3 (3.1%) AE related Mim8, n (%) 0 4 (19.0%) 1 (5.0%) 0 20 (20.4%) 0 17 (17.3) AE leading to Mim8 d/c,a n (%) 0 1 (4.8%) 0 0 2 (2.0%) 0 0
- 53. NXT007 IgG = immunoglobulin G. Teranishi-Ikawa et al, 2024; NCT05987449. IgG anti-FIXa/anti-FX bispecific antibody 30-40x higher affinity than emicizumab Activity similar to emicizumab but at 30-fold lower concentration Phase 1/2 trial recruiting adolescent and adults with moderate/severe HA with or without inhibitors Potential near-normal hemostatic state
- 54. Slide courtesy of Guy Young, MD. XI XIa Ca++ IX IXa Ca++ X Xa Ca+ + VIIIa VIII VII VIIa TF Xa X Ca++ Prothrombin (II) Thrombin (IIa) Fibrinogen (I) Fibrin XIII XIIIa Cross-linked fibrin V Va Thrombin (IIa) Antithrombin Protein C Protein Ca Thrombin (IIa) Protein S Protein S Thrombin (IIa) Ca++ Tissue factor pathway inhibitor (TFPI) Emicizumab MIM8 NXT007 Concizumab Marstacimab MG1113 Fitusiran Serpin PC Anti-APC Mab Anti-PS Mab Anti-PS siRNA Anti-PS Mab Anti-PS siRNA Efanesoctocog alfa Novel Therapeutics Mechanisms of Action Inhibits Replacement therapy Substitution therapy Rebalancing agents
- 55. Tissue Factor Pathway Inhibitor (TFPI) Chowdary, 2018. TFP I Tissue factor + FVIIa FIXa (+ FVIIa) FXa (+ FVa) FIIa (thrombin) Fibrinogen Fibrin X Anti-TFPI antibody against K2 domain Anti-TFPI antibody against K1 and K2 domains Anti-TFPI TFPI TFPI VIIa TF X Xa VIIa TF X Xa VIIa TF X Xa
- 56. Marstacimab IgG1 = immunoglobulin G1; FXa = factor Xa. Kwak et al, 2020. Monoclonal Antibody Targeted Against TFPI TFPI-K2 Marstacimab-HC Marstacimab-LC R107 Marstacimab is a human IgG1 monoclonal antibody against TFPI that targeting the K2 domain, blocking its ability to inhibit FXa and preventing the binding of the K1 domain to FVIIa
- 57. Once-Weekly Marstacimab-hncq HympavziTM et al, 2024. First and only subcutaneous prophylactic treatment in hemophilia A or B without inhibitors that comes in a fixed-dose, prefilled pen Marstacimab-hncq is indicated for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adult and pediatric patients 12 years of age and older with: Hemophilia A (congenital factor VIII deficiency) without factor VIII inhibitors Hemophilia B (congenital factor IX deficiency) without factor IX inhibitors Marstacimab-hncq Injection 150 mg/mL
- 58. BASIS: Efficacy and Safety of Marstacimab ATP = active treatment phase. Matino et al, 2023; Mahlangu, 2025. On-demand treatment with FVIII/FIX Current SOC (n=37) Routine prophylaxis with FVIII/FIX Current SOC (n=91) With FVIII/FIX inhibitors (ongoing) Male patients aged ≥12 years and ≥35 kg with severe hem A or hem B Dose escalation of marstacimab (optional) ~45-day screening 6-month observation N=128 12-month open-label active treatment N=116 Following the 12-month ATP, eligible patients could opt-in to participate in an open-label extension study Open-label extension N=87 Without FVIII/FIX inhibitors n=128 Marstacimab prophylaxis Single SC loading dose of 300 mg followed by QW 150 mg Marstacimab prophylaxis continued
- 59. BASIS: Marstacimab (cont.) OD = on-demand; RP = routine prophylaxis. Acharya et al, 2024; NCT03938792. Eligibility Criteria for Non-Inhibitor Cohort • Patients with a history of coronary artery disease, venous or arterial thrombosis, or ischemic disease were excluded from the study • Scheduled surgery during study period • Platelet count <100,000/µL • Hemoglobin level <10 g/dL • Current RP with bypassing agent, noncoagulation nonfactor replacement therapy, or any previous gene therapy product • Current immunomodulatory drugs • Severe hem A (FVIII <1%) without inhibitors or severe hem B (FIX ≤2%) without inhibitors • Male, ≥12 to <75 years of age, minimum weight of 35 kg at screening • No detection or history of inhibitors against FVIII or FIX • OD group: ≥6 acute bleeding episodes (spontaneous or traumatic) that required coagulation factor infusion during the 6-month period prior to enrollment • RP group: ≥80% compliance with FVIII/FIX regimen 6 months prior to enrollment Select Inclusion Criteria Select Exclusion Criteria
- 60. BASIS: Marstacimab (cont.) OP = observational phase. Matino et al, 2023. On-Demand (OD) Arm Marstacimab during ATP (n=33) Superior vs On-Demand 50 40 30 20 10 0 Mean ABR for treated bleeds (95% CI) 38.00 (95% CI: 31.03, 46.54) On-demand during OP (n=33) 3.18 (95% CI: 2.09, 4.85) 92% Reduction P<0.0001 12-month ATP 6-month OP Median ABR 35.73 2.02
- 61. BASIS: Marstacimab (cont.) Matino et al, 2023. Routine Prophylaxis Arm Marstacimab during ATP (n=83) Non-Inferior vs Routine Prophylaxis 15 10 5 0 Mean ABR for treated bleeds (95% CI) 7.85 (95% CI: 5.09, 10.61) Routine prophylaxis during OP (n=83) 5.08 (95% CI: 3.40, 6.77) 35% Reduction 12-month ATP 6-month OP Median ABR 2.16 2.02
- 62. Anti-Drug Antibodies ADAs including NAbs had no identified clinically significant effect on safety or efficacy of marstacimab over the treatment duration of 12 months 23/116 patients tested positive for ADAs (19.8%) NAb development was low at 6/23 (26.1%) BASIS: Marstacimab (cont.) PI = prescribing information; ADAs = anti-drug antibodies; NAbs = neutralizing antibodies. Matino et al, 2023 Safety Adverse Reaction Number of Patients (N=116), n (%) Injection site reaction 11 (9%) Headache 8 (7%) Pruritus 4 (3%) In the BASIS study, the majority of adverse reactions were mild-to-moderate and there were no reported discontinuations due to treatment-related adverse reactions. See PI for guidance on when to discontinue marstacimab. Adverse Reactions Reported in ≥3% of Patients Treated With Marstacimab During the BASIS Trial 12-Month Active Treatment Phase
- 63. Concizumab-mtci Alhemo® prescribing information, 2025. Concizumab-mtci is a tissue factor pathway inhibitor (TFPI)– antagonist indicated for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adult and pediatric patients 12 years of age and older with: Hemophilia A with or without FVIII inhibitors Hemophilia B with or without FIX inhibitors Concizumab-mtci 150 mg/1.5 mL (100 mg/mL)
- 65. Mean ABR was 1.7 and median ABR was 0, and 64% of participants who received concizumab (Arm 2, n=33) had 0 treated bleeds at 24 weeks 9.8 (6.5; 20.2) 0.0 (0.0; 3.3) 0 2 4 6 8 10 12 14 No prophylaxis (n=19; arm 1) Concizumab prophylaxis* (n=114; arms 2–4) Median ABR (IQR) Concizumab prophylaxis (arm 2; n=33) No prophylaxis (arm 1; n=19) 11.8 (7.0; 19.9) 1.7 (1.0; 2.9) 0 2 4 6 8 10 12 14 Mean ABR (95% CI) explorer7: Concizumab aIncludes participants previously on demand that were randomized to receive concizumab prophylaxis (Arm 2. n=33), participants that transferred from the explorer4 trial, and an additional group of participants that were on prior prophylaxis or on demand (Arms 3 and 4, respe ctively, n=81). IQR = interquartile range. Jiménez Yuste et al, 2022; Mathias et al, 2023. Phase 3 Efficacy Data ABR at Primary Analysis Cutoffa in People With Hemophilia A or B With Inhibitors
- 66. Treated spontaneous and traumatic bleeding episodes HAwI No PPX (Arm 1; n=9) HAwI Concizumab PPX (Arm 2; n=18) ABR ratio (95% CI) HBwI No PPX (Arm 1; n=10) HBwI Concizumab PPX (Arm 2; n=15) ABR ratio (95% CI) Estimated mean ABR 18.3 (10.2-32.9) 1.6 (0.9-2.8) 0.09 (0.04-0.18) 7.2 (2.6-20.1) 2.2 (0.8-6.5) 0.31 (0.07-1.36) Treated spontaneous and traumatic bleeding episodes HAwI Concizumab PPX (Arms 1-4; n=76) HBwI Concizumab PPX (Arms 1-4; n=51) Overall median ABR (IQR) 0.0 (0.0-3.7) 0.0 (0.0-3.3) Number of episodes 128 152 explorer7: Concizumab (cont.) HAwI = hemophilia A with inhibitors; HBwI = hemophilia B with inhibitors. Frei-Jones et al, 2022; Jiménez Yuste et al, 2022. Phase 3 Efficacy Data Efficacy results were consistent when split by hemophilia subtype
- 67. explorer7: Concizumab (cont.) aIncludes Arm 1 participants who started concizumab treatment in the extension part of the trial, and Arm 2-4 participants. Jiménez Yuste et al, 2022. Phase 3 Efficacy Data n (%) No Prophylaxis Concizumab Prophylaxis (Arm 2) Concizumab Prophylaxis (All Participantsa) Total events 8 (42.1%) 20 (60.6%) 80 (63.0%) Serious events 3 (15.8%) 6 (18.2%) 14 (11.0%) Fatal events 1 (5.3%) 2 (6.1%) 2 (1.6%) Drug withdrawn 0 2 (6.1%) 4 (3.1%) Thromboembolic events 0 1 (3.0%) 1 (0.8%) Thromboembolic events after restart 0 0 0 Hypersensitivity-type reaction 0 1 (3.0%) 2 (1.6%) Injection-site reaction 0 6 (18.2%) 26 (20.5%)
- 68. Estimated Treatment Difference for Hemo-TEM Scores explorer7: Concizumab (cont.) CZM = concizumab; ETD = estimated treatment difference; Hemo-TEM = Hemophilia Treatment Experience Measure; PPX = prophylaxis. Hampton et al, 2023. HRQOL Was Improved With Concizumab in the Phase 3 explorer7 Study
- 69. Concizumab Phase 3 Trials SD = standard deviation. Windyga et al, 2024. Efficacy at 56-Week Cutoff explorer7 explorer8 HAwI HBwI HA HB No. patients in full analysis set 76 51 80 64 Cumulative years of exposure in data analysis set 84.3 56.2 111.9 71.7 Treated Spontaneous and Traumatic Bleeding Episodes Median ABR (IQR) 0.7 (0.0-3.0) 1.1 (0.0-3.2) 1.7 (0.0-4.5) 2.8 (0.0-6.4) Mean ABR (SD) 2.1 (4.1) 4.3 (11.5) 3.9 (6.6) 6.4 (14.2) Min; max ABR 0.0; 31.8 0.0; 66.4 0.0; 37.1 0.0; 91.3
- 70. Concizumab Phase 3 Trials (cont.) aSafety analysis set. PYE = patient-years of exposure. Windyga et al, 2024. Efficacy at 56-Week Cutoff explorer7: HAwI/HBwI (n=127a) explorer8: HA/HB (n=151a) n (%) No. of Events Events/ PYE n (%) No. of Events Events/PYE Total AE 91 (71.7%) 467 2.9 118 (78.1%) 528 2.7 Serious AE 22 (17.3%) 27 0.2 20 (13.2%) 30 0.2 Fatal AE 4 (3.1) 6 0.0 1 (0.7%) 1 0.0 AE leading to drug withdrawal 4 (3.1%) 4 0.0 6 (4.0%) 8 0 TE prior to tx pause 1 (0.8%) 1 0.0 2 (1.3%) 4 0.0 TE from tx restart to 56-wk cutoff 0.0 0.0 - 0 - - Hypersensitivity 2 (1.6%) 2 0.0 0 - - Injection site reactions 30 (23.6%) 60 0.4 26 (17.2%) 46 0.2 Medication errors 4 (3.1%) 6 0.0 4 (2.6%) 5 0.0 • Fatal events: – explorer7: COVID-19 (n=1), intracranial hemorrhage (n=1), alcoholic coma (n=1), and 1 patient with fatal humerus fracture, femur fracture, and road traffic accident – explorer8: intra-abdominal hemorrhage (n=1)
- 71. Novel Therapeutics Mechanisms of Action Slide courtesy of Guy Young, MD. XI XIa Ca++ IX IXa Ca++ X Xa Ca+ + VIIIa VIII VII VIIa TF Xa X Ca++ Prothrombin (II) Thrombin (IIa) Fibrinogen (I) Fibrin XIII XIIIa Cross-linked fibrin V Va Thrombin (IIa) Antithrombin Protein C Protein Ca Thrombin (IIa) Protein S Protein S Thrombin (IIa) Ca++ Tissue factor pathway inhibitor (TFPI) Emicizumab MIM8 NXT007 Concizumab Marstacimab MG1113 Fitusiran Serpin PC Anti-APC Mab Anti-PS Mab Anti-PS siRNA Anti-PS Mab Anti-PS siRNA Efanesoctocog alfa Inhibits Replacement therapy Substitution therapy Rebalancing agents
- 72. Targeting Antithrombin: Fitusiran Young, Srivastava et al, 2023; Srivastava et al, 2023; Kenet et al, 2024; NCT03974113; NCT03754790. Small interference RNA → degradation of mRNA Monthly subcutaneous injection ATLAS Clinical Trial Program
- 74. Fitusiran BPA = bypassing agent; CPC = clotting factor concentrate; ALT = alanine aminotransferase; AST =aspartate aminotransferase; ULN = upper limit of normal. Young, Srivastava et al, 2023; Srivastava et al, 2023; Kenet et al, 2022. Study population People with haemophilia A or B aged ≥12 years with inhibitors (N=57) received fitusiran 80 mg prophylaxis or on-demand BPAs People with haemophilia A or B aged ≥12 years without inhibitors (N=120) received fitusiran 80 mg prophylaxis or on-demand CFCs People with haemophilia A or B aged ≥12 years with or without inhibitors (N=80) with prior BPA/CFC prophylaxis received fitusiran 80 mg prophylaxis Efficacy Safety 91% reduction in bleeding rates compared with on-demand BPAs (P<0.0001); median ABR was 0.0 90% reduction in bleeding rates compared with on-demand CFCs (P<0.0001); median ABR was 0.0 61% reduction in bleeding rates compared with prior BPA/CFC prophylaxis (P=0.0008); median ABR was 0.0 Completed Phase 3 Trials Thrombotic events: 2 participants (5.0%) ALT/AST elevations 3x ULN: 10 participants (24.0%) Thrombotic events: none ALT/AST elevations 3x ULN: 15 participants (19.0%) Thrombotic events: 2 participants (3.0%) ALT/AST elevations 3x ULN: 17 participants (25.4%) Mean AT levels (SD) 10.7% (2.9) 11.8% (4.0) 11.0% (2.5) Fitusiran has been studied in the largest pre-approval hemophilia cohort to date, with 335 participants exposed from 24 countries
- 75. Revised AT-Based Dosing Regimen QM = every month; SS = steady state; AT = antithrombin. Pipe et al, 2022; Young, Lenting, et al, 2023; Young et al, 2024. Designed to Enhance the Benefit-Risk Profile of Fitusiran >1 AT level <15% ≥2 SS AT levels >35% ≥2 SS AT levels >35% ≥2 SS AT levels >35% >1 AT level <15% Revised AT-based dosing regimen targeting an AT level of 15%-35% Original dose regimen 80 mg QM Regular AT monitoring was used to guide dosing Lower-dose cohort for participants requiring <50 mg Q2M 20 mg QM 80 mg QM 20 mg Q2M 50 mg Q2M 50 mg QM Discontinue
- 76. Approved AT-Based Dose Modification Qftlia prescribing information, 2025. Initial dose: 50 mg Q2M Measure AT activity at Month 1, 3, 5, and 6 following the starting dose and after any dose modification Last Dosage Administered Antithrombin Activity Level Dose Modification 50 mg Q2M <15% 20 mg Q2M 15%-35% Continue current dosage >35% after 6 months 50 mg Q1M 20 mg Q2M <15% 10 mg Q2M 15%-35% Continue current dosage >35% after 6 months 20 mg Q1M 10 mg Q2M <15% Discontinue fitusiran 15%-35% Continue current dosage >35% after 6 months 10 mg every month
- 77. Revised AT-Based Dosing Regimen (cont.) a80 participants (38%) required 0 and 119 (56%) required 1 dose change to achieve AT range 15%-35%. Pipe et al, 2022; Young, Lenting et al, 2023; Young et al, 2024. Achieved Its Aim of Maintaining AT Levels Between 15%-35% AT value by visit during primary efficacy period • 78% (n=167) were on Q2M regimens at final dose • 94% (n=199) required 0 or 1 dose adjustment to achieve AT 15%-35% Mean (SD) AT level was 23.5% (4.6) with the AT-DR 0 10 20 30 40 50 60 Month 7 Month 8 Month 9 Month 10 Month 11 Month 12 Month 13 Study Visit Antithrombin level (%)
- 78. Thrombotic Events AT-DR = antithrombin-based dose regimen; ODR = original dose regimen; TEAESI = thrombotic event adverse event of special interest; TE = thrombotic event. Young et al, 2025. TEAESIs, n (%) AT-Based Dose Regimen (n=286) Original Dose Regimen (n=270) Thrombotic events 4 9 Participants with any thrombotic events (%) 4 (1.4) 7 (2.6) Exposure adjusted incidence rate (per 100 patient years) 0.82 2.28 Based on a recently submitted abstract on Optum claims database analysis, the rate of TEs with the AT-DR are similar to the general hemophilia population 2.28 0.82 0 0.5 1 1.5 2 2.5 3 3.5 4 4.5 5 Original dose regimen (N=270) AT-based dose regimen (N=286) Exposure adjusted incidence rate (per 100 patient-years) Incidence of thrombotic events Incidence of Thrombotic Events Was Reduced With AT-DR vs ODR
- 79. Young et al, 2025. TEAESIs, n (%) AT-Based Dose Regimen (n=286) Original Dose Regimen (n=270) Cholecystitis/Cholelithiasis Participants with any Cholecystitis/Cholelithiasis 11 (3.8%) 45 (16.7%) Exposure adjusted incidence rate (per 100 patient years) 2.26 14.67 TEAESIs, n (%) AT-Based Dose Regimen (n=286) Original Dose Regimen (n=270) ALT/AST elevations > 3xULN Participants with any ALT/AST elevations >3x ULN 10 (3.5%) 56 (20.7%) Exposure adjusted incidence rate (per 100 patient years) 2.06 18.25 Majority of ALT or AST elevations 3x ULN with the AT-DR were transient and reported as recovered/resolved, with a median of 212 and 186 days to onset and median time to normalisation of 88 and 49 days, respectively ALT/AST Elevations and Cholecystitis/Cholelithiasis Incidence of ALT/AST elevations 3x ULN and cholecystitis/cholelithiasis was substantially reduced with AT-DR vs ODR
- 80. Young et al, 2025. 1.9 (0.0; 5.6) 3.8 (0.0; 11.2) 3.7 (0.0; 7.5) 0 2 4 6 8 10 Inhibitor (n=69) Non-inhibitor (n=128) Overall (N=197) Median ABR (IQR) Observed median ABR with the fitusiran AT-DR Proportion of participants with 0 or 1 bleed in the primary efficacy period: 31.5% 37.7% 28.1% 0 Bleeds 47.2% 53.6% 43.7% 0-1 Bleeds 80 Fitusiran AT-DR provided clinically meaningful bleed control in participants with hemophilia A and B with and without inhibitors ATLAS-OLE: Fitusiran AT-DR
- 81. Properties of Rebalancing Agents Young et al, 2024; Alhemo® prescribing information, 2025; HympavziTM et al, 2024; Matino et al, 2025. MOA Drug Dosing Regimen Development Phase Comments Anti-AT siRNA Fitusiran SC monthly or every other month Approved Thrombotic events led to a new dosing regimen targeting AT levels between 15%-35% Anti-TFPI monoclonal antibodies Concizumab SC daily Approved Dose to be adjusted based on concizumab levels Marstacimab SC weekly Approved Dosing does not require adjustment
- 82. Patient Case: Hemophilia A (cont.) 14-year-old male with severe hemophilia A with history of transient inhibitor Baseline FVIII <1% Initially started on SHL FVIII Trial of emicizumab for 12 months; developed a large muscle bleed but wants to know his options
- 83. Polling Question What is known about muscle bleeds in patients on emicizumab? a. Emicizumab does not prevent muscle bleeds b. Emicizumab is only effective against joint bleeds c. There are reports of serious muscle bleeds especially in teenagers and young adults on emicizumab d. Factor concentrates are proven to be more effective against muscle bleeds that emicizumab
- 84. Polling Question Which of the following options could be considered for this patient? a. Efanesoctocog alfa (though this would mean going from a subcutaneous drug to an intravenously administered drug) b. Switch to fitusiran c. Switch to concizumab d. Switch to marstacimab e. All of the above
- 85. Patient Case: Hemophilia B 19-year-old male with severe hemophilia B without history of inhibitor Baseline FIX <1% Initially started on SHL FIX product 3 times a week Recently attempted rFIX:Fc weekly with good trough at Day 7 (2%) and ABR~3 Active in soccer (promises doesn’t head the soccer ball) Wants to consider alternative therapy as he is getting “tired” of frequent injections
- 86. Polling Question What are the novel options for prophylaxis for a young adult with severe hemophilia B? a. Factor replacement (SHL and EHL products) are the only option b. An anti-TFPI subcutaneous drug such as concizumab or marstacimab c. An siRNA subcutaneous option such as fitusiran monthly or every 2 months d. Options b and c are reasonable for prophylaxis
- 87. Strategies for Advanced Therapies in Hemophilia LV = lentiviral gene therapy; MSC = mesenchymal stem cell; CRISPR = clustered regularly interspaced short palindromic repeats; ZFN = zinc-finger nuclease. Rodríguez-Merchán et al, 2021. Advanced Therapies Gene Therapy Cell Therapy IN VIVO AAVs LVs EX VIVO MSCs CRIPSR ZFN (AAVs)
- 89. FVIII Activity (mITT Population) CSA = chromogenic substrate assay; mITT = modified intention-to-treat; Q = quartile; SE = standard error. Leavitt et al, 2025. Nearly Stable Compared to Year 4 Week 260 Mean ± SE: 13.7 ± 2.1 IU/dL Median (Q1, Q3): 6.2 (2.4, 14.2) IU/dL Week 208 Mean ± SE: 15.9 ± 2.5 IU/dL Median (Q1, Q3): 6.5 (2.8, 17.7) IU/dL Week 156 Mean ± SE: 18.4 ± 2.7 IU/dL Median (Q1, Q3): 8.3 (3.0, 17.2) IU/dL Mean ± SE FVIII activity (IU/dL) 0 20 40 60 80 100 Study week 4 28 52 76 104 128 156 180 208 232 260 mITT population n = 132 132 132 132 132 132 132 132 132 132 132 For participants who discontinued the study, missing FVIII values post-discontinuation were imputed as 0 IU/dL through the data cutoff date.
- 90. Valoctocogene Roxaparvovec (cont.) CI = confidence interval. Leavitt et al, 2025. Annualized Rate of Treated Bleeding Episodes 0 5 10 15 Mean + SD Median (Q3) 83.3% reduction Change in mean, −4.02 (95% CI, −5.24 to −2.80); P<0.0001 112 112 110 110 112 108 n= 112 2.8 0 0 0 0 0 0.2 4.8 0.9 0.7 1.0 0.9 0.6 0.8 ABR (bleeds/year) Baseline Year 1 Year 2 Year 3 Year 4 Year 5 All post- prophylaxis
- 91. Valoctocogene Roxaparvovec (cont.) Leavitt et al, 2025. Annualized Rate of Factor VIII Use Baseline Year 1 Year 2 Year 3 Year 4 Year 5 All post- prophylaxis 0 50 100 150 200 250 128.6 0 0 0 0 0 0.8 135.9 1.5 3.4 8.6 10.7 10.6 6.9 Change in mean, −129.0 (95% CI, −138.6 to −119.3); P<0.0001 112 112 110 110 112 108 n = 112 94.9% reduction AFR (infusions/year) Mean + SD Median + Q3
- 92. Participants, n (%) Year 1 (N=134) Year 2 (N=134) Year 3 (N=132) Year 4 (N=131) Year 5 (N=129) All Follow-Up AEs 134 (100.0%) 112 (83.6%) 104 (78.8%) 98 (74.8%) 102 (79.1%) 134 (100.0%) SAEs 21 (15.7%) 6 (4.5%) 9 (6.8%) 11 (8.4%) 4 (3.1%) 37 (27.6%) Treatment-related AEsa 124 (92.5%) 27 (20.1%) 15 (11.5%) 10 (7.6%) 5 (3.9%) 124 (92.5%) Glucocorticoid-related AEsa 81 (60.4%) 10 (7.5%) 1 (0.8%) 1 (0.8%) 0 82 (61.2%) AEs of special interest ALT elevation 116 (86.6%) 39 (29.1%) 31 (23.7%) 49 (37.4%) 52 (40.3%) 125 (93.3%) ALT elevation grade ≥3 10 (7.5%) 1 (0.7%) 0 0 0 10 (7.5%) Potential Hy’s law case 0 0 0 0 0 0 Infusion-related reactionsb 12 (9.0%) 0 0 0 0 12 (9.0%) Systemic hypersensitivity 7 (5.2%) 0 0 0 0 7 (5.2%) Anaphylactic or anaphylactoid reactions 3 (2.2%) 0 0 0 0 3 (2.2%) Thromboembolic events 0 0 0 0 0 0 Anti-FVIII neutralizing antibodies 0 0 0 0 0 0 Malignancy (except nonmelanoma skin cancer) 0 0 1 (0.8%) 0 0 1 (0.7%) Valoctogene Roxaparvovec (cont.) aTreatment-related and glucocorticoid-related adverse events (AEs) were assessed by the investigator. bInfusion-related reactions were defined as AEs occurring during valoctocogene roxaparvovec infusion or within 6 hours post-infusion. ITT = intention-to-treat; SAE = serious AE. Leavitt et al, 2025. Safety
- 93. Gene Therapy to Deliver Factor IX Pipe, 2025. Etranacogene Dezaparvovec
- 94. Etranacogene Dezaparvovec Pipe, 2025. Endogenous Factor IX Activity Over 18 Months After Treatment
- 95. Etranacogene Dezaparvovec (cont.) Pipe, 2025. Annualized Bleeding Rate 60% (31/54) of participants experienced no joint bleeds over 0-4 years post-treatment
- 96. Etranacogene Dezaparvovec (cont.) Pipe, 2025. Changes in Factor Consumption and Number of Infusions Of 54 participants treated with etranacogene dezaparvovec: • 2 participants remained on continuous FIX prophylaxis post-infusion (high NAb) titer of 3,212, n=1; ~10% of the dose, n=1 • 94.4% (51/54) participants discontinued continuous FIX prophylaxis at 4 years • 1 participant returned to continuous FIX prophylaxis 30 months following infusion 46.3% (25/54) of participants received no FIX infusion over the 4-year period post-treatment infusions
- 97. aPlanned as only 1 infusion total. Srivastava et al, 2020; Hemlibra® prescribing information, 2025; Hermans et al, 2024; Shima et al, 2025; Young et al, 2024; Alhemo® prescribing information, 2025; Hympavzi et al, 2024; Matino et al, 2025; Mahlangu et al, 2023; Pipe et al, 2023. Drug/MOA Infusions Per Year Comments Factor replacement 52-183 Only IV. Other administration methods have been tried but have not worked well Emi/Mim8/NXT007 13-52 Very long washout (months) with no antidote Fitusiran 6-12 Very long washout (months) but antidote (AT infusion) is available Concizumab 365 Daily infusion, but advantage of rapid washout No antidote Marstacimab 52 No antidote Gene therapy 1a No ability to reverse effects for better or worse Novel Agents by Infusions Per Year
- 98. So, How Do We Choose?
- 99. SHARE Decision Making Model Dennison Himmelfarb et al, 2023.
- 100. Patient Categories Age Hemophilia type Severity Inhibitor status <18 years >18 years Hem A Hem B Sev Mod Mild Positive Negative Sev = severe; Mod = moderate. Srivastava et al, 2020. Patient Factors When Choosing Therapy
- 101. Patient Categories Age Hemophilia type Severity Inhibitor status <18 years >18 years Hem A Hem B Sev Mod Mild Positive Negative Patient Categories Venous access Adherence Risk-averse Lifestyle (work or play) Good Poor Good Bad No Med Yes Higher-risk job/active Sedentary Patient Factors When Choosing Therapy Srivastava et al, 2020.
- 102. Patient Categories Age Hemophilia type Severity Inhibitor status <18 years >18 years Hem A Hem B Sev Mod Mild Positive Negative Patient Categories Venous access Adherence Risk-averse Lifestyle (work or play) Good Poor Good Bad No Med Yes Higher-risk job/active Sedentary Patient Categories Age Cardiovascular risk factors Individual patient values <65 >65 Yes No High efficacy vs low treatment burden Patient Factors When Choosing Therapy Srivastava et al, 2020.
- 103. Patient Case: Hemophilia A (cont.) 14-year-old male with severe hemophilia A with history of transient inhibitor Baseline FVIII <1% Initially started on SHL FVIII Trial of emicizumab for 12 months; developed a large muscle bleed but wants to know his options
- 104. Polling Question Which of the following options could be considered for this patient? a. Efanesoctocog alfa (though this would mean going from a subcutaneous drug to an intravenously administered drug) b. Switch to fitusiran c. Switch to concizumab d. Switch to marstacimab e. All of the above
- 105. Polling Question Which of the following options could be considered for this patient? a. Efanesoctocog alfa (though this would mean going from a subcutaneous drug to an intravenously administered drug) b. Switch to fitusiran c. Switch to concizumab d. Switch to marstacimab e. All of the above
- 106. Patient Case: Hemophilia A (cont.) 14-year-old male with severe hemophilia A with history of transient inhibitor Additional history is that he is very active in sports, including soccer and baseball, and he believes his muscle bleed occurred while playing soccer Continuing these sports is very important for him He places the most value on a different therapy with one that can provide him the highest protection from bleeding and is not concerned as much about the treatment burden
- 107. Polling Question Given the additional history, which of the following options would you choose? a. Efanesoctocog alfa b. Switch to fitusiran c. Switch to concizumab d. Switch to marstacimab e. All of the above
- 108. Polling Question Given the additional history, which of the following options would you choose? a. Efanesoctocog alfa b. Switch to fitusiran c. Switch to concizumab d. Switch to marstacimab e. All of the above
- 109. Patient Case: Hemophilia A (cont.) Efanesoctocog alfa provides factor levels ranging from a peak of ~100% to a trough of ~15%-20% Emicizumab has a FVIII equivalence of about 10%-20%, and that level is steady (no peaks and troughs) It is not yet clear what the FVIII equivalence of fitusiran, concizumab, or marstacimab are, though bleed protection (granted across different trials with no head-to-head comparison) seems similar to emicizumab Given all the options, this patient’s high value on participation in sports means that his highest FVIII protection would come from receiving efanesoctocog alfa
- 110. Patient Case: Hemophilia B (cont.) 19-year-old male with severe hemophilia B without history of inhibitor Baseline FIX <1% Initially started on SHL FIX product 3 times a week Recently attempted rFIX:Fc weekly with good trough at Day 7 (2%) and ABR~3 Active in soccer (promises doesn’t head the soccer ball) Wants to consider alternative therapy as getting “tired” of frequent injections
- 111. Polling Question FP = recombinant human albumin; N9:GP = nonacog beta pegol. Which of the following options could be considered for this patient? a. Switch to FIX:FP product or N9:GP product b. Switch to concizumab c. Switch to fitusiran d. Switch to marstacimab e. All of the above
- 112. Polling Question FP = recombinant human albumin; N9:GP = nonacog beta pegol. Which of the following options could be considered for this patient? a. Switch to FIX:FP product or N9:GP product b. Switch to concizumab c. Switch to fitusiran d. Switch to marstacimab e. All of the above
- 113. Patient Case: Hemophilia B (cont.) 19-year-old male with severe hemophilia B without history of inhibitor Baseline FIX <1% On EHL FIX product currently and desires a change to a non-IV product He and his family chose marstacimab Weekly administration, single subcutaneous injection
- 114. Patient Case: Hemophilia B (cont.) 19-year-old male with severe hemophilia B without history of inhibitor He and his family chose marstacimab Weekly administration, single subcutaneous injection He self injects weekly on the weekend Feels more independent as he is in control of his meds Day 1: loading dose 300 mg SC injection Day 8: start once-weekly maintenance dose 150 mg SC injection once weekly
- 115. Key Takeaways Over the past 3 years, 6 new medications have been approved for hemophilia A and B—these include a novel factor therapy, 3 nonfactor therapies, and 2 gene therapies Each has their pros and cons, and hemophilia treaters must familiarize themselves with the properties, dosing regimen, laboratory monitoring, and safety and efficacy for all of them Through shared decision making, the best therapy for each individual patient’s situation can be selected
- 116. References Acharya SS, Matino D, Palladino A, et al (2024). Safety and efficacy of the anti-tissue factor pathway inhibitor marstacimab in participants with severe hemophilia without inhibitors: results from the phase 3 BASIS trial and ongoing long-term extension study. Presented at the Thrombosis & Hemostasis Summit 2024. Abstract 206. Alhemo® (concizumab-mtci) prescribing information. Novo Nordisk. Available at: accessdata.fda.gov/scripts/cder/daf/index. Bakeer N & Shapiro AD (2019). Merging into the mainstream: the evolution of the role of point-of-care musculoskeletal ultrasound in hemophilia. F1000Res, 8:F10000 Faculty Rev-1029 [Version 1]. DOI:10.12688/f1000research.16039.1 Batsuli G, Wheeler AP, Weyand AC (2023). Severe muscle bleeds in children and young adults with hemophilia A on emicizumab prophylaxis: real-world retrospective multi- institutional cohort. Am J Hematol, 98(10):285-287. DOI:10.1002/ajh.27039 Boulden Warren B, Thornhill D, Stein J, et al (2020). Young adult outcomes of childhood prophylaxis for severe hemophilia A: results of the Joint Outcome Continuation Study. Blood Adv, 4(11):2451-2459. DOI:10.1182/bloodadvances.2019001311 Calcaterra I, Iannuzzo G, Dell’Aquila F & Dario Di Minno MN (2020). Pathophysiological role of synovitis in hemophilic arthropathy development: a two-hit hypothesis. Front Physiol, 11:541. DOI:10.3389/fphys.2020.00541 Callaghan MU, Negrier C, Paz-Priel I, et al (2021). Long-term outcomes emicizumab prophylaxis for hemophilia A with or without FVIII inhibitors from the HAVEN 1-4 studies. Blood, 137(16):2231-2242. DOI:10.1182/blood.2020009217 Callaghan MU, Sidonio R & Pipe SW (2018). Novel therapeutics for hemophilia and other bleeding disorders. Blood, 132(1):23-30. DOI:10.1182/blood-2017-09-743385 Chowdary P (2018). Inhibition of tissue factor pathway inhibitor (TFPI) as a treatment for haemophilia: rationale with focus on concizumab. Drugs, 78:881-890. DOI:10.1007/s40265-018-0922-6 Clinicaltrials.gov (2024). Long-term safety and efficacy of fitusiran in patients with hemophilia A or B, with or without inhibitory antibodies to Factor VIII or IX (ATLAS-OLE). NLM identifier: NCT03753790 Clinicaltrials.gov (2025). A study to evaluate the safety, tolerability, pharmacokinetics, pharmacodynamics, and efficacy of NXT007 in persons with severe or moderate hemophilia A. NLM identifier: NCT05987449 Clinicaltrials.gov (2025). Fitusiran prophylaxis in male pediatric subjects aged 1 to less than 12 years with hemophilia A or B (ATLAS-PEDS). NLM Identifier: NCT03974113. Clinicaltrials.gov (2025). Study of the efficacy and safety of PF-06741086 in adult and teenage patients with severe hemophilia A or moderately severe to severe hemophilia B. NLM identifier: NCT03938792
- 117. References (cont.) Dennison Himmelfarb CR, Beckie TM, Allen LA, et al (2023). Shared decision-making and cardiovascular health: a scientific statement from the American Heart Association. Circulation, 148(11):912-931. DOI:10.1161/CIR.0000000000001162 Foppen W, van der Schaff IC, Beek FJA, et al (2018). Diagnostic accuracy of point-of-care ultrasound for evaluation of early blood-induced joint changes: comparison with MRI. Haemophilia, 24(6):971-979. DOI:10.1111/hae.13524 Frei-Jones M, Cepo K, d’Oiron R, et al (2022). Subcutaneous prophylaxis in patients with hemophilia A or B with inhibitors: efficacy and safety results by hemophilia subtype from the Phase 3 explorer 7 trial. Blood, 140(suppl_1):466-468. DOI:10.1182/blood-2022-166522 Gamal Andrawes N, Fayek MH, El-Din N & Mostafa RA (2020). Effect of low-dose factor VIII prophylaxis therapy on bone mineral density and 25(OH) vitamin D level in children with severe haemophilia A, Haemophilia, 26(2):325-332. DOI:10.1111/hae.13917 GlobeNewswire.com (2025). Novo Nordisk A/S: once-weekly Mim8 is well-tolerated and efficacious in children living with haemophilia A with and without inhibitors. Available at:https://www.globenewswire.com/news-release/2025/02/07/3022568/0/en/Novo-Nordisk-A-S-Once-weekly-Mim8-is-well-tolerated-and-efficacious-in-children-living- with-haemophilia-A-with-and-without-inhibitors.html Gouw SC, Timmer MA, Srivastava A, et al (2019). Measurement of joint health in persons with haemophilia: a systematic review of the measurement properties of haemophilia- specific instruments. Haemophilia, 25(1):e1-e10. DOI:10.1111/hae.13631 Hampton K, Knoebl P, Neergaard JS, et al (2023). Treatment burden and patient preference in patients with haemophilia A or B with inhibitors on concizumab propylaxis: results from the phase 3 explorer7 study. Haemophilia, 29(suppl_1). 16th Annual Congress of European Association for Haemophilia and Allied Disorders 2023. Abstract: PO104 Hemlibra® prescribing information (emicizumab-kxwh) injection (2025). Genentech, Inc. Available at: accessdata.fda.gov/scripts/cder/daf/index.cfm Hermans C, Ay C, Escuriola C, et al (2024). Mim8 prophylaxis beyond bleeding: investigating multifaceted, patient-reported outcomes for hemophilia A in the FRONTIER2 study. Blood, 144(suppl_1):1212. DOI:10.1182/blood-2024-198395 HympavziTM (Marstacimab-hncq) injection prescribing information (2024). Pfizer Inc. Available at: accessdata.fda.gov/scripts/cder/daf/index.cfm Jiménez Yuste, Angchaisuksiri P, Castaman G, et al (2022). Concizumab prophylaxis in patients with haemophilia A or B with inhibitors: efficacy and safety result from the primary analysis of the Phase 3 explorer7 trial. Presented at the ISTH 2022 Congress. Abstract LV01.2
- 118. References (cont.) Kenet G, Nolan B, Zulfikar B, et al (2024). Fitusiran prophylaxis in people with hemophilia A or B who switched from prior BPA/CFC prophylaxis: the ATLAS-PPX trial. Blood, 143(22):2256-2269. DOI:10.1182/blood.2023021864 Kilzilocak H & Young G (2019). Diagnosis and treatment of hemophilia. Clin Adv Hematol Oncol, 17(6):344-351. Kitazawa T, Igawa T, Sampei Z, et al (2012). A bispecific antibody to factors IXa and X restores factor VIII hemostatic activity in a hemophilia A model. Nature Medicine, 18:1570- 1574. DOI:10.1038/nm.2942 Konkle BA, Shapiro AD, Quon DV, et al (2020). BIV001 fusion protein as factor VIII replacement therapy for hemophilia A. N Engl J Med, 383(11):1018-1027. DOI:10.1056/NEJMoa2002699 Kwak H, Lee S, Jo S, et al (2020). MG1113, a specific anti-tissue factor pathway inhibitor antibody, rebalances the coagulation system and promotes hemostasis in hemophilia. Research and Practice on Thrombosis and Haemostasis, 4(8):1301-1312. DOI:10.1002/rth2.12438 Leavitt AD, Mahlangu J, Raheja P, et al (2025). Final GENEr8-1 results confirm enduring efficacy, safety, and quality of life improvements 5 years after valoctogene roxaparvovec gene transfer. Presented at the International Society on Thrombosis and Haemostasis (ISTH) Annual Congress 2025. Abstract: #PB0804 Lillicrap D, Lund J, Jensen K, et al (2024). FVIII in vitro bioequivalence of Mim8 haemostatic effect by thrombin generation assays [oral presentation]. Presented at ISTH 2024. Abstract OC 50.5. Lissitchkov T, Willemze A, Katragadda S, et al (2022). Efanesoctocog alfa for hemophilia A: results from a phase 1 repeat-dose study. Blood Adv, 6(4):1089-1094. DOI:10.1182/bloodadvances.2021006119 Mahlangu J (2025). Marsacimab for the treatment of hemophilia A or B. Biologics, 19:379-386. DOI:10.2147/BTT.S500480 Manco-Johnson MJ, Abshire TC, Shapiro A, et al (2007). Prophylaxis versus episoic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med, 357(6):535- 544. DOI:10.1056/NEJMoa067659 Mancuso ME, Matsushita T, Chowdary P, et al (2024). Efficacy and safety of Mim8 prophylaxis in adults and adolescents with hemophelia A with or without inhibitors: Phase 3, open-label, randomized, controlled FRONTIER2 study. Presented at the ISTH 2024 Congress. Abstract LB 01.5. Mathias M, Linari S, Bruzelius M, et al (2024). Longer-term concizumab prophylaxis in patients with haemophilia A or B with inhibitor: ≥1-year (56-week cut-off) efficacy and safety results from the phase 3 explorer7 trial. Haemophilia, 29(suppl_1). Presented at the 16th Annual Congress of European Association for Haemophilia and Allied Disorders 2023. Abstract PO140 Matino D, Acharya S, Palladino A, et al (2023). Efficacy and safety of the anti-tissue factor pathway inhibitor marstacimab in participangs with severe hemophilia without inhibitors: results from the phase 3 BASIS trial. Blood, 142(suppl_1):285. DOI:10.1182/blood-2023-181263 Matino D, Palladiono A, Taylor CT, et al (2025). Marstacimab prophylaxis in hemophilia A/B without inhibitors: results from the phase 3 BASIS trial. Blood, [Online ahead of print]. Blood. 2024027468. DOI:10.1182/blood.2024027468
- 119. References (cont.) Matsushita T, Shapiro A, Abraham A, et al (2023). Phase 3 trial of concizumab in hemophilia with inhibitors. N Engl J Med, 389(9):783-794. DOI:10.1056/NEJMoa2216455 Miesbach W, Klamroth R, Oldenburg J & Tiede A (2022). Gene therapy for hemophilia – opportunities and risks. Dtsch Arztebl Int, 119(51-52):887-894. DOI:10.3238/arztebl.m2022.0353 Østergaard H, Lund J, Griesen PJ, et al (2021). A factor VIIIa-mimetic bispecific antibody, Mim8, ameliorates bleeding upon severe vascular challenge in hemophilia A mice. Blood, 138(14):1258-1268. DOI:10.1182/blood.2020010331 Ozelo MC & Yamaguti-Hayakawa GG (2022). Impact of novel hemophilia therapies around the world. Res Pract Thromb Haemost, 6(3):12695. DOI:10.1002/rth2.12695 Pipe SW (2025). The phase 3 HOPE-B trial shows 4-year durability of sustained near-normal FIX activity, bleed protection and favourable safety in adults with severe or moderately severe haemophilia B [oral presentation]. Presented at 18th Annual Congress of the European Association for Haemophila and Allied Disorders. Abstract OR14 Pipe SW, Leebeek FWG, Recht M, et al (2023). Gene therapy with etranacogene dezaparvovec for hemophilia B. N Engl J Med, 388(8):706-718. DOI:10.1056/NEJMoa2211644 Pipe SW, Srivastava A, Klamroth R, et al (2022). Fitusiran, an investigational siRNA therapeutic targeting antithrombin: analysis of antithrombin levels and thrombin generation from a phase 3 study in people with hemophilia A or B without inhibitors [oral presentation]. Presented at the ISTH Congress 2022. Abstract OC50.2 QfitliaTM (fitusiran) injection prescribing information (2025). Genzyme Corporation. Available at: accessdata.fda.gov/scripts/cder/daf/index.cfm Rodríguez-Merchán C, De Pablo-Moreno JA & Liras A (2021). Gene therapy in hemophilia: recent advances. Int J Mol Sci, 22(14):7647. DOI:10.3390/ijms22147647 Shima M, You CW, Park YS (2025). NXT007 prophylaxis in emicizumab-naïve persons with hemophilia A without inhibitor: Phase I/II study [Oral presentation]. ISTH 2025 Congress. Abstract OC 20.3. Srivastava A, Rangarajan S, Kavakli K, et al (2023). Fitusiran prophylaxis in people with severe haemophilia A or haemophiia B without inhibitors (ATLAS-A/B): a multicentre, open-label, randomised, phase 3 trial. Lancet Haematol, 10(5):322-332. DOI:10.1016/S2352-3026(23)00037-6 Srivastava A, Santagostino E, Dougall A, et al (2020). WFH guidelines for the management of hemophilia, 3rd edition. Haemophilia, 26(suppl_6):1-158. DOI:10.1111/hae.14046 St-Louis J, Abad A, Funk S, et al (2022). The hemophilia joint health score version 2.1 validation in adult patients study: a multicenter international study. Res Pract Thromb Haemost, 6(2):e12690. DOI:10.1002/rth2.12690 Teranishi-Ikawa Y, Soeda T, Koga H, et al (2023). A bispecific antibody NXT007 exerts a hemostatic activity in hemophilia A monkeys enough to keep a nonhemophilic state. J Thromb Haemost, 22:430-440. DOI:10.1016/j.jtha.2023.09.034 von Drygalski A, Chowdary P, Kulkarni R, et al (2023). Efanesoctocog alfa prophylaxis for patients with severe hemophilia A. N Engl J Med, 388(4):310-318. DOI:10.1056/NEJMoa2209226
- 120. References (cont.) Wilkins RA, Stephensen D, Siddle H, et al (2022). Twelve-month prevalence of hemoarthrosis and joint disease using the Haemophilia Joint Health score: evaluation of the UK National Haemophilia Database and Haemtrack patient reported data: an observational study. BMJ Open, 12:e052358. DOI:10.1136/bmjopen-2021-052358 Windyga J, Apte S, Astermark J, et al (2024). Efficacy and safety of concizumab prophylaxis in haemophilia A or B with and without inhibitors: 56-week cut-off results of the phase 3 explorer7 and explorer8 studies. Presented at the. 17th annual Congress of the European Association for Haemophilia and Allied Disorders 2024. Abstract OR07. Young G (2023). Nonfactor therapies for hemophilia. Hemasphere, 7(6):911. DOI:10.1097/HS9.0000000000000911 Young G, Avakli K, Pipe SW, et al (2024). Safety and efficacy of the fitusiran revised antithrombin-based dose regimen in people with haemophilia A or B, with or without inhibitors (ATLAS-OLE). Hematology, Transfusion, and Cell Therapy, 46(suppl_4):S564-565. DOI:10.1016/j.htct.2024.09.948 Young G, Kavakli K, Klamroth R, et al (2025). Safety and efficacy of a fitusiran antithrombin-based dose regimen in people with hemophilia A or B, with or without inhibitors: the ATLAS-OLE study. Blood, 145(25):2966-2977. DOI: 10.1182/blood.2024027008 Young G, Lenting PJ, Croteau SE (2023). Antithrombin lowering in hemophilia: a closer look at fitusiran. Res Pract Thromb Haemost, 7(4):100179. DOI:10.1016/j.rpth.2023.100179 Young G, Srivastava A, Kavakl K, et al (2023). Efficacy and safety of fitusiran prophylaxis in people with hemophilia B with inhibitors (ATLAS-INH): a multicentre, open-label, randomised phase 3 trial. Lancet, 401(10386):1427-1437. DOI:10.1016/S0140-6736(23)00284-2



















































































































![References
Acharya SS, Matino D, Palladino A, et al (2024). Safety and efficacy of the anti-tissue factor pathway inhibitor marstacimab in participants with severe hemophilia without
inhibitors: results from the phase 3 BASIS trial and ongoing long-term extension study. Presented at the Thrombosis & Hemostasis Summit 2024. Abstract 206.
Alhemo® (concizumab-mtci) prescribing information. Novo Nordisk. Available at: accessdata.fda.gov/scripts/cder/daf/index.
Bakeer N & Shapiro AD (2019). Merging into the mainstream: the evolution of the role of point-of-care musculoskeletal ultrasound in hemophilia. F1000Res, 8:F10000 Faculty
Rev-1029 [Version 1]. DOI:10.12688/f1000research.16039.1
Batsuli G, Wheeler AP, Weyand AC (2023). Severe muscle bleeds in children and young adults with hemophilia A on emicizumab prophylaxis: real-world retrospective multi-
institutional cohort. Am J Hematol, 98(10):285-287. DOI:10.1002/ajh.27039
Boulden Warren B, Thornhill D, Stein J, et al (2020). Young adult outcomes of childhood prophylaxis for severe hemophilia A: results of the Joint Outcome Continuation Study.
Blood Adv, 4(11):2451-2459. DOI:10.1182/bloodadvances.2019001311
Calcaterra I, Iannuzzo G, Dell’Aquila F & Dario Di Minno MN (2020). Pathophysiological role of synovitis in hemophilic arthropathy development: a two-hit hypothesis. Front
Physiol, 11:541. DOI:10.3389/fphys.2020.00541
Callaghan MU, Negrier C, Paz-Priel I, et al (2021). Long-term outcomes emicizumab prophylaxis for hemophilia A with or without FVIII inhibitors from the HAVEN 1-4 studies.
Blood, 137(16):2231-2242. DOI:10.1182/blood.2020009217
Callaghan MU, Sidonio R & Pipe SW (2018). Novel therapeutics for hemophilia and other bleeding disorders. Blood, 132(1):23-30. DOI:10.1182/blood-2017-09-743385
Chowdary P (2018). Inhibition of tissue factor pathway inhibitor (TFPI) as a treatment for haemophilia: rationale with focus on concizumab. Drugs, 78:881-890.
DOI:10.1007/s40265-018-0922-6
Clinicaltrials.gov (2024). Long-term safety and efficacy of fitusiran in patients with hemophilia A or B, with or without inhibitory antibodies to Factor VIII or IX (ATLAS-OLE). NLM
identifier: NCT03753790
Clinicaltrials.gov (2025). A study to evaluate the safety, tolerability, pharmacokinetics, pharmacodynamics, and efficacy of NXT007 in persons with severe or moderate hemophilia
A. NLM identifier: NCT05987449
Clinicaltrials.gov (2025). Fitusiran prophylaxis in male pediatric subjects aged 1 to less than 12 years with hemophilia A or B (ATLAS-PEDS). NLM Identifier: NCT03974113.
Clinicaltrials.gov (2025). Study of the efficacy and safety of PF-06741086 in adult and teenage patients with severe hemophilia A or moderately severe to severe hemophilia B.
NLM identifier: NCT03938792](https://image.slidesharecdn.com/247hemepresentationforslideshare-250819203432-120952f7/85/Raising-the-Bar-for-Hemophilia-Management-116-320.jpg)

![References (cont.)
Kenet G, Nolan B, Zulfikar B, et al (2024). Fitusiran prophylaxis in people with hemophilia A or B who switched from prior BPA/CFC prophylaxis: the ATLAS-PPX trial. Blood,
143(22):2256-2269. DOI:10.1182/blood.2023021864
Kilzilocak H & Young G (2019). Diagnosis and treatment of hemophilia. Clin Adv Hematol Oncol, 17(6):344-351.
Kitazawa T, Igawa T, Sampei Z, et al (2012). A bispecific antibody to factors IXa and X restores factor VIII hemostatic activity in a hemophilia A model. Nature Medicine, 18:1570-
1574. DOI:10.1038/nm.2942
Konkle BA, Shapiro AD, Quon DV, et al (2020). BIV001 fusion protein as factor VIII replacement therapy for hemophilia A. N Engl J Med, 383(11):1018-1027.
DOI:10.1056/NEJMoa2002699
Kwak H, Lee S, Jo S, et al (2020). MG1113, a specific anti-tissue factor pathway inhibitor antibody, rebalances the coagulation system and promotes hemostasis in hemophilia.
Research and Practice on Thrombosis and Haemostasis, 4(8):1301-1312. DOI:10.1002/rth2.12438
Leavitt AD, Mahlangu J, Raheja P, et al (2025). Final GENEr8-1 results confirm enduring efficacy, safety, and quality of life improvements 5 years after valoctogene roxaparvovec
gene transfer. Presented at the International Society on Thrombosis and Haemostasis (ISTH) Annual Congress 2025. Abstract: #PB0804
Lillicrap D, Lund J, Jensen K, et al (2024). FVIII in vitro bioequivalence of Mim8 haemostatic effect by thrombin generation assays [oral presentation]. Presented at ISTH 2024.
Abstract OC 50.5.
Lissitchkov T, Willemze A, Katragadda S, et al (2022). Efanesoctocog alfa for hemophilia A: results from a phase 1 repeat-dose study. Blood Adv, 6(4):1089-1094.
DOI:10.1182/bloodadvances.2021006119
Mahlangu J (2025). Marsacimab for the treatment of hemophilia A or B. Biologics, 19:379-386. DOI:10.2147/BTT.S500480
Manco-Johnson MJ, Abshire TC, Shapiro A, et al (2007). Prophylaxis versus episoic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med, 357(6):535-
544. DOI:10.1056/NEJMoa067659
Mancuso ME, Matsushita T, Chowdary P, et al (2024). Efficacy and safety of Mim8 prophylaxis in adults and adolescents with hemophelia A with or without inhibitors: Phase 3,
open-label, randomized, controlled FRONTIER2 study. Presented at the ISTH 2024 Congress. Abstract LB 01.5.
Mathias M, Linari S, Bruzelius M, et al (2024). Longer-term concizumab prophylaxis in patients with haemophilia A or B with inhibitor: ≥1-year (56-week cut-off) efficacy and
safety results from the phase 3 explorer7 trial. Haemophilia, 29(suppl_1). Presented at the 16th Annual Congress of European Association for Haemophilia and Allied
Disorders 2023. Abstract PO140
Matino D, Acharya S, Palladino A, et al (2023). Efficacy and safety of the anti-tissue factor pathway inhibitor marstacimab in participangs with severe hemophilia without
inhibitors: results from the phase 3 BASIS trial. Blood, 142(suppl_1):285. DOI:10.1182/blood-2023-181263
Matino D, Palladiono A, Taylor CT, et al (2025). Marstacimab prophylaxis in hemophilia A/B without inhibitors: results from the phase 3 BASIS trial. Blood, [Online ahead of print].
Blood. 2024027468. DOI:10.1182/blood.2024027468](https://image.slidesharecdn.com/247hemepresentationforslideshare-250819203432-120952f7/85/Raising-the-Bar-for-Hemophilia-Management-118-320.jpg)
![References (cont.)
Matsushita T, Shapiro A, Abraham A, et al (2023). Phase 3 trial of concizumab in hemophilia with inhibitors. N Engl J Med, 389(9):783-794. DOI:10.1056/NEJMoa2216455
Miesbach W, Klamroth R, Oldenburg J & Tiede A (2022). Gene therapy for hemophilia – opportunities and risks. Dtsch Arztebl Int, 119(51-52):887-894.
DOI:10.3238/arztebl.m2022.0353
Østergaard H, Lund J, Griesen PJ, et al (2021). A factor VIIIa-mimetic bispecific antibody, Mim8, ameliorates bleeding upon severe vascular challenge in hemophilia A mice. Blood,
138(14):1258-1268. DOI:10.1182/blood.2020010331
Ozelo MC & Yamaguti-Hayakawa GG (2022). Impact of novel hemophilia therapies around the world. Res Pract Thromb Haemost, 6(3):12695. DOI:10.1002/rth2.12695
Pipe SW (2025). The phase 3 HOPE-B trial shows 4-year durability of sustained near-normal FIX activity, bleed protection and favourable safety in adults with severe or moderately
severe haemophilia B [oral presentation]. Presented at 18th Annual Congress of the European Association for Haemophila and Allied Disorders. Abstract OR14
Pipe SW, Leebeek FWG, Recht M, et al (2023). Gene therapy with etranacogene dezaparvovec for hemophilia B. N Engl J Med, 388(8):706-718. DOI:10.1056/NEJMoa2211644
Pipe SW, Srivastava A, Klamroth R, et al (2022). Fitusiran, an investigational siRNA therapeutic targeting antithrombin: analysis of antithrombin levels and thrombin generation
from a phase 3 study in people with hemophilia A or B without inhibitors [oral presentation]. Presented at the ISTH Congress 2022. Abstract OC50.2
QfitliaTM (fitusiran) injection prescribing information (2025). Genzyme Corporation. Available at: accessdata.fda.gov/scripts/cder/daf/index.cfm
Rodríguez-Merchán C, De Pablo-Moreno JA & Liras A (2021). Gene therapy in hemophilia: recent advances. Int J Mol Sci, 22(14):7647. DOI:10.3390/ijms22147647
Shima M, You CW, Park YS (2025). NXT007 prophylaxis in emicizumab-naïve persons with hemophilia A without inhibitor: Phase I/II study [Oral presentation]. ISTH 2025
Congress. Abstract OC 20.3.
Srivastava A, Rangarajan S, Kavakli K, et al (2023). Fitusiran prophylaxis in people with severe haemophilia A or haemophiia B without inhibitors (ATLAS-A/B): a multicentre,
open-label, randomised, phase 3 trial. Lancet Haematol, 10(5):322-332. DOI:10.1016/S2352-3026(23)00037-6
Srivastava A, Santagostino E, Dougall A, et al (2020). WFH guidelines for the management of hemophilia, 3rd edition. Haemophilia, 26(suppl_6):1-158. DOI:10.1111/hae.14046
St-Louis J, Abad A, Funk S, et al (2022). The hemophilia joint health score version 2.1 validation in adult patients study: a multicenter international study. Res Pract Thromb
Haemost, 6(2):e12690. DOI:10.1002/rth2.12690
Teranishi-Ikawa Y, Soeda T, Koga H, et al (2023). A bispecific antibody NXT007 exerts a hemostatic activity in hemophilia A monkeys enough to keep a nonhemophilic state. J
Thromb Haemost, 22:430-440. DOI:10.1016/j.jtha.2023.09.034
von Drygalski A, Chowdary P, Kulkarni R, et al (2023). Efanesoctocog alfa prophylaxis for patients with severe hemophilia A. N Engl J Med, 388(4):310-318.
DOI:10.1056/NEJMoa2209226](https://image.slidesharecdn.com/247hemepresentationforslideshare-250819203432-120952f7/85/Raising-the-Bar-for-Hemophilia-Management-119-320.jpg)
