Ca ovary staging etiology pathogenesis
- 2. Each ovary is 2.5–5.0 cm long, 1.5–3.0 cm thick, and 0.7–1.5 cm wide and is attached by its mesenteric attachment to the broad ligament, the mesovarium. The infundibular pelvic ligament, which includes the ovarian artery and vein is present laterally while the ovarian ligament is present medially. The blood vessels enter the hilus of the ovary.
- 3. The germinal epithelium lines the outside, or the capsule of the ovary. The inner medulla of the ovary is a fibromuscular layer with blood vessels. The cortex, including the stroma, consists of follicles, corpus luteum, and corpus albicans.
- 5. Blood Supply: Arterial blood supply to the ovaries is via the ovarian arteries, which have their origin in the anterior surface of the aorta, inferior to the renal vessels. The left ovarian vein empties into the left renal vein, which then enters into the vena cava, and the right ovarian vein empties into the vena cava inferior to the right renal vein.
- 6. The lymphatic drainage of the ovary follows its blood supply through the infundibulopelvic ligament to nodes in the para-aortic region. Lymphatic drainage through the broad ligament and parametrial channels can also result in involvement of pelvic sidewall lymphatics, including the external iliac, obturator, and hypogastric chains. Spread may rarely occur along the course of the round ligament, resulting in involvement of inguinal lymph nodes.
- 7. INTRODUCTION Ovarian neoplasms consist of several histopathologic entities; treatment depends on the specific tumor type. Epithelial ovarian cancer comprises the majority of malignant ovarian neoplasms (about 90%); less common histopathologies, including - ovarian low malignant potential [LMP] tumor, - malignant germ cell neoplasms, - carcinosarcomas (malignant mixed Müllerian tumors [MMMTs] and - malignant sex cord- stromal tumors.
- 8. Epithelial ovarian cancer typically occurs in postmenopausal women & mostly present in advanced stage. Most germ cell tumors present in younger women, and sex cord–stromal tumors may occur at any age. In contrast, other types of ovarian cancer such as germ cell and sex cord–stromal tumors are often localized in distribution, more amenable to surgical resection, and have a more favorable prognosis.
- 9. EPIDEMIOLOGY Both in the United States and in Europe, ovarian cancer is the fifth most common cause of cancer death. African American women in the United States have a lower incidence of ovarian cancer (10.3 per 100,000 women) compared to white women, The lifetime risk of developing sporadic epithelial ovarian cancer is approximately 1.7%, although patients with a familial predisposition have a much higher lifetime risk, in the range of 10% to 40%. The median age at diagnosis for sporadic disease is 60 years, although patients with a genetic predisposition may develop this tumor earlier, often in their fifth decade.
- 10. RISK FACTORS Established risk factors for ovarian cancer include a strong family history, early menarche, late menopause, increasing age, and nulliparity.
- 11. REPRODUCTIVE FACTORS: PREGNANCY AND BREAST FEEDING Early menarche and late menopause increase the risk of ovarian cancer. while increased parity, breastfeeding, and the use of oral contraceptives (OCP) reduce the risk. These findings suggest that the risk for ovarian cancer is linked to the number of ovulations that a woman experiences throughout her lifetime. Breast-feeding probably reduces ovarian cancer incidence through several mechanisms, including suppression of ovulation, reduced serum concentrations of estradiol and LH, and elevated FSH levels.
- 12. ORAL CONTRACEPTIVES Women who use OCP for at least 5 years reduce their risk of ovarian cancer by an average of 50%, and the level of protection increases with duration of use. Therefore, protection against ovarian cancer is probably the most important non-contraceptive benefit of OCPs.
- 13. HORMONE REPLACEMENT THERAPY a review of the major studies suggests a 30% to 58% increase in ovarian cancer risk in women who take HRT. The risk of ovarian cancer was found to increase with duration of HRT exposure, and the largest risk was seen among women who used HRT for over 20 years. Most studies show that a treatment duration of less than 5 years carries no statistically increased ovarian cancer risk.
- 14. SURGERY: TUBAL INTERRUPTION AND HYSTERECTOMY In general, studies have confirmed that both tubal interruption and hysterectomy at least partially protect against the development of ovarian cancer. Possible explanations for the protective effect of tubal interruption against ovarian cancer include - an impaired blood supply to the ovaries/distal tubes through the superior branch of the uterine artery leading in most women to earlier menopause (and fewer lifetime ovulations) - the possibility that occluding the tube blocks the upward flow of carcinogens from the uterus and reduces pelvic infection rates.
- 15. SMOKING,OBESITY Smoking is not generally considered a risk factor for ovarian cancer. However, current smoking seems to be associated with an increased risk of developing a mucinous ovarian cancer. Smoking cessation reduces this risk back to baseline over 20 years. Obesity does not appear to be associated with the most aggressive types of ovarian cancer
- 16. PATHOGENESIS AND PATTERNS OF METASTASES Epithelial ovarian neoplasms are thought to arise from the surface epithelium covering the ovary, which is contiguous with peritoneal mesothelium. However, emerging evidence suggests that a subset of epithelial ovarian cancers may instead originate in the fallopian tube fimbria, subsequently spreading to the ovary or peritoneal cavity. Germ cell tumors most likely originate in cells derived from the primitive streak that ultimately migrated to the gonads. The ovarian stroma consists of granulosa cells, theca cells, and fibroblasts, which give rise to the sex cord–stromal tumors.
- 17. Mutation in the p53 protooncogene occurs in over 50% of cases, predominantly involving tumors in patients with advanced stage and high-grade serous histology. In contrast, mutations in B-raf, K-ras, PTEN, or β-catenin may be seen in endometrioid, mucinous, or low-grade histologies . Amplification of the HER2/neu gene is observed in only approximately 8% of patients and confers a poorer prognosis.
- 18. Overexpression of pro-apoptotic genes such as BAX is associated with chemoresponsiveness and a more favorable prognosis. Surface adhesion proteins such as CD44H and β-1 integrins have been shown to mediate transperitoneal spread of this tumor by promoting the attachment of cancer cells to the peritoneal mesothelial lining.
- 19. The tumor typically spreads to the omentum and to peritoneal surfaces such as the underside of the diaphragm, paracolic gutters, and bowel serosa. The lymphatic drainage of the ovary follows its blood supply through the infundibulopelvic ligament to nodes in the para- aortic region.
- 20. Lymphatic drainage through the broad ligament and parametrial channels can also result in involvement of pelvic sidewall lymphatics, including the external iliac, obturator, and hypogastric chains. Spread may rarely occur along the course of the round ligament, resulting in involvement of inguinal lymph nodes. Approximately 10% to 15% of patients with ovarian cancer that appears to be localized to the ovaries have metastases to para-aortic lymph nodes, and retroperitoneal lymph node involvement is found in over 50% of patients with advanced disease.
- 21. The most common site of extra-abdominal spread is the pleural space (thought to occur via transdiaphragmatic lymphatics), where it causes a malignant pleural effusion in some patients. Hematogenous metastases to the liver, spleen, or lung can also occur during the course of the disease, but are relatively uncommon at presentation. Bone or central nervous system metastases may rarely be observed in patients who have lived for many years after initial diagnosis, during which unusual patterns of disease spread may occur.
- 22. WHO CLASSIFICATION OF OVARIAN TUMORS The three main histologic types, which include nearly all ovarian cancers, are epithelial tumors, sex-cord stromal tumors, and germ cell tumors.
- 23. I. EPITHELIAL TUMORS a. Serous tumors 1. Benign serous cystadenoma 2. Of borderline malignancy: (carcinomas of low potential malignancy) 3. Serous cystadenocarcinoma b. Mucinous tumors 1. Benign mucinous cystadenoma 2. Of borderline malignancy: (carcinomas of low potential malignancy) 3. Mucinous cystadenocarcinoma c. Endometrioid tumors 1. Benign endometrioid cystadenoma 2. (carcinomas of low potential malignancy) 3. Endometrioid adenocarcinoma
- 24. d. Clear cell tumors 1. Benign clear cell tumors 2 (low potential malignancy) 3. Clear cell cystadenocarcinoma e. Brenner (transitional cell tumors) 1. Benign Brenner 2. Borderline malignancy 3. Malignant 4. Transitional cell f. Squamous cell tumor g. Undifferentiated carcinoma 1. A malignant tumor of epithelial structure that is too poorly differentiated to be placed in any other group h. Mixed epithelial tumor 1. Tumors composed of two or more of the five major cell types of common epithelial tumors (types should be specifi ed)
- 25. SEX CORD- STROMAL TUMORS Granulosa – Stromal Cell Tumor Granulosa cell tumor Tumor in thecoma- fibroma group Fibroma Unclassified Androblastoma: Sertoli- Leydig Cell Tumor Well differentiated Tubular androblastoma Sertoli- leydig cell tumor Hilus cell tumor of intermedite differentiation Poorly differentiated( sarcomatoid) Gyandroblastoma Lipid cell tumors Unclassified
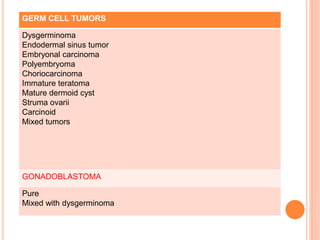
- 26. GERM CELL TUMORS Dysgerminoma Endodermal sinus tumor Embryonal carcinoma Polyembryoma Choriocarcinoma Immature teratoma Mature dermoid cyst Struma ovarii Carcinoid Mixed tumors GONADOBLASTOMA Pure Mixed with dysgerminoma
- 27. Histology Features Papillary serous The most common type of epithelial ovarian cancer. May contain psammoma bodies and is often associated with CA 125 elevation. Endometrioid Sometimes associated with endometriosis or an independent uterine cancer of similar histology. May occur with early stage disease in younger patients, although advanced disease is also possible. Mucinous May rarely be associated with pseudomyxoma peritoneii. CA 125 levels may not be markedly elevated. Relatively chemoresistant. Differential diagnosis of a mucinous ovarian tumor includes metastatic disease from an appendiceal primary. Clear cell The most chemoresistant type of ovarian cancer. Sometimes associated with endometriosis or humorally mediated hypercalcemia. Table 104.2. Common Histologic Types of Epithelial Ovarian Cancer
- 28. CLINICAL PRESENTATION AND DIAGNOSTIC WORKUP Clinical Presentation of Patients with a Benign Adnexal Mass or Early-Stage Ovarian Cancer Almost all patients with small adnexal tumors are asymptomatic, and the mass is usually discovered incidentally during a work-up for other conditions. With increasing size, adnexal masses cause pelvic pressure and pain by compressing surrounding structures. A larger pelvic tumor can cause genitourinary symptoms including urgency, frequency, and dyspareunia.
- 29. Posteriorly, a fixed pelvic tumor compresses the sigmoid colon, causing severe constipation. it is impossible to differentiate a benign from a malignant ovarian mass by clinical examination alone. An ovarian mass becomes a surgical emergency if a patient with an adnexal mass has sudden onset of abdominal pain. The differential includes torsion and possible infarction of the ovarian mass.
- 30. vaginal bleeding in a patient with an adnexal mass could be an indication of synchronous endometrial and ovarian cancers or a granulosa cell tumor that produces estrogen, causing abnormal bleeding. Sertoli-Leydig cell tumors can lead to virilization.
- 31. CLINICAL PRESENTATION OF PATIENTS WITH ADVANCED OVARIAN CANCER General, nonspecific symptoms associated with advanced ovarian cancer include anorexia, fatigue, early satiety and loss of appetite. bloating and diffuse, dull, constant abdominal pain caused by the infiltration of the peritoneum and the bowel mesentery, or by extensive ascites. While weight loss is unusual, tumor cachexia can be a presenting sign in patients with high-volume disease and long standing partial bowel obstruction.
- 32. If the tumor has metastasized to the omentum, patients complain of upper abdominal discomfort with nausea, belching, early satiety and fullness. In addition, ascites can cause dyspnea. Signs of bowel obstruction, severe urinary symptoms, intense pelvic pain, and ascites are likely to indicate miliary dissemination on the peritoneal surfaces and large-volume advanced disease.
- 33. CLINICAL EXMINATION A focused clinical exam should begin with an assessment of supraclavicular lymph nodes, a breast exam, and percussion of the lungs to detect a pleural effusion. The involvement of the umbilicus by an ovarian cancer is called “Sister Mary Joseph nodule” Examine for the presence of ascites, the size and mobility of the adnexal mass, and the presence of enlarged inguinal lymph nodes.
- 34. The bimanual and rectovaginal exam should attempt to evaluate and characterize the adnexal mass in respect to size, borders (smooth/irregular), mobility (fixed/mobile), and location. Invasive ovarian cancers often have irregular borders and are often fixed to the pelvic sidewall and fill the cul-the-sac.
- 35. LABORATORY TESTS The laboratory work-up for patients with suspected ovarian cancer generally includes Complete blood count. LFTs RFTs
- 36. The CA 125 serum level is elevated in more than 80% of serous epithelial ovarian cancers. it can also be elevated in a variety of benign gynecologic conditions (such as endometriosis, pelvic inflammatory disease, or pregnancy) and nongynecologic malignancies (such as breast, lung, and gastrointestinal cancers). Furthermore, the CA 125 level is elevated in only approximately 50% of patients with early stage epithelial ovarian cancer, which also limits its value as a screening test.
- 37. Other tumor markers, such as CA 19-9, which is elevated in some mucinous ovarian carcinomas, and carcinoembryonic antigen (CEA) are less frequently useful. Postoperatively, the CA 125 level provides a sensitive way to monitor treatment response and development of disease recurrence.
- 38. It is best to avoid percutaneous biopsy during the initial evaluation, which can result in cyst rupture and spillage of malignant cells into the peritoneal cavity.
- 39. IMAGING OF ADNEXAL MASSES, EARLY AND ADVANCED OVARIAN CANCER Ultrasound First modality to diagnose or confirm the clinically suspected adnexal mass. Ultrasound can detect and characterize ovarian size and morphology. TVS is better than trans abdominal approach for assessment of morphological features.
- 40. Benign tumors often appear on ultrasound as unilateral with smooth walls, with a few cysts, no solid elements and an absence of ascites. Functional cysts have thin walls and are fluid filled. Dermoids are often cystic with hyper-echoic areas (teeth).
- 41. TVU is more sensitive at detecting ovarian tumors compared to other tests such as computed tomography (CT), and it can provide qualitative information about the mass that might suggest malignancy. The classic sonographic finding of malignancy is a “complex” cyst, -containing both solid and cystic components, -with septations and -internal echogenicity, -irregular wall, -mural nodules.
- 42. Color Doppler imaging evaluates blood flow to an ovarian mass and can potentially identify a malignant process based on the presence of abnormal neovascularization & low resistance flow pattern. Positive predictive value of 98% has been reported for transvaginal colour doppler.
- 45. Chest radiographs may sometimes be performed to evaluate the presence of pleural effusions, which occur in 10% of patients with epithelial ovarian cancer at diagnosis.
- 46. COMPUTED TOMOGRAPHY/CT CT features of ovarian malignancy - a multilocular cyst with thick internal septations, - solid mural nodules, - complex solid-cystic mass or - lobulated papillary mass. Solid components enhanced well with iv contrast. In the pelvis, a CT scan allows for characterization of the adnexal mass and any involvement of surrounding organs (bladder, sigmoid, ureter, pelvic sidewall involvement). In the upper abdomen retroperitoneal adenopathy, omental involvement, and intrahepatic liver involvement can be detected reliably by CT scans with intravenous contrast.
- 48. MAGNETIC RESONANCE IMAGING—MRI Morphological features of ovarian cancer on MRI are similar to CECT , but because of excellent soft tissue contrast, the details are better demonstrated. Contrast enhancement helps to differentiate solid & necrotic areas. MRI is better than other modalities in determining the origin of pelvic mass. Also accurate in differentiate b/w benign and malignant mass.
- 49. MRI features favouring malignant mass - size > 4 cm, - large solid component - wall or septal thickness> 3mm - nodularity or vegetations - necrosis. And ancillary features.
- 50. In recurrent cancer, MRI is especially useful for differentiating between postsurgical changes and a recurrence on the vaginal dome, small bowel mesentery, splenic hilum, liver surface, or diaphragm.
- 51. SCREENING AND EARLY DETECTION A successful screening test for ovarian cancer should be capable of identifying the majority of patients with precancerous lesions or early disease. Because a positive screening test for ovarian cancer would result in major surgery with associated morbidity, costs, and even mortality. -the false-positive rate must be low, and -positive predictive value (PPV) relatively high (at least 10%). At present, there are no screening tests for epithelial ovarian cancer that convincingly meet these criteria. The ability of tumor cells to exfoliate from the ovarian surface and to spread in an asymptomatic fashion impedes the development of successful screening approaches that would allow for early diagnosis.
- 52. The CA 125 serum level is not a useful screening test when used alone, since elevations are not specific for ovarian cancer. Elevation may be observed in cirrhosis, peritonitis, pleuritis, pancreatitis, endometriosis, uterine leiomyoma, benign ovarian cysts, and pelvic inflammatory disease. Elevated in other malignancies such as breast, lung, colorectal, pancreatic, and gastric cancers.
- 53. More recently, a number of candidate markers have been discovered that show promise for enhancing the accuracy of CA 125 levels, such as - HE4 (human epididymis 4), - Osteopontin, - Mesothelin, and - Osteoblast-stimulating factor-2. Levels of OVX-1 and macrophage colony-stimulating factor have been found to be elevated in patients with clinically evident ovarian cancer but normal CA 125 levels, which suggests that these markers may be complementary to CA 125. None of these tests has been proven to have sufficient sensitivity and specificity for routine screening at the current time.
- 54. Measurement of the CA 125 level has been combined with performance of TVU in an attempt to improve screening. Early studies of TVU suggested a sensitivity of close to 100% but a specificity of 98%, which is insufficient to achieve a PPV of 10%.
- 55. SCREENING TRIALS In the United States, the Prostate, Lung, Colorectal, Ovarian Cancer Screening Trial uses measurement of CA 125 level (single threshold elevation of >35 U/mL) and TVU together, performed annually, as a first-line screen. If either test is positive, the woman is referred for surgical consultation. no mortality reduction was observed for patients randomized to the screening arm.
- 56. An ongoing trial is assessing screening for ovarian cancer (UK Collaborative Trial of Ovarian Cancer Screening [UKCTOCS]) using multimodality screening with ultrasound and CA-125 versus either ultrasound alone or no screening. Preliminary results suggest that multimodality screening is more effective at detecting early-stage cancer.
- 57. An earlier study by Jacobs et al. in which 21,935 average-risk postmenopausal women were assigned to undergo three annual screenings or no screening. The screening protocol used CA 125 level as a first- line screen and referred the patient for TVU if the CA 125 level was >30 U/ml. If the TVU revealed an ovarian mass, the patient was referred for surgical consultation. Statistically significant improvement in survival. average of 72.9 months in the screening group and 41.8 months in the control group.
- 58. The OVA1 test uses 5 markers (including transthyretin, apolipoprotein A1, transferrin, beta-2 microglobulin, and CA-125). The Society of Gynecologic Oncology (SGO), the FDA, and the Mayo Clinic have stated that the OVA1 test should not be used as a screening tool to detect ovarian cancer.
- 59. HEREDITARY OVARIAN CARCINOMA Approximately 5% to 10% of patients with epithelial ovarian carcinoma carry a germline mutation that places them at substantially increased risk of developing this disease. The breast–ovarian cancer syndrome accounts for approximately 90% of hereditary ovarian cancer and is often suspected whenever - the pedigree reveals multiple affected family members with ovarian cancer, - bilateral or early onset breast cancer, - both breast and ovarian cancer in the same individual, or - a male relative with breast cancer.
- 60. The high incidence of breast and ovarian cancers in these families is due to inherited germline mutations in the BRCA1 or BRCA2 genes. The BRCA1 gene, located on chromosome band 17q12-21, and the BRCA2 gene, located on chromosome band 13q12- 13, were identified and linked to hereditary breast and ovarian cancers in the 1990s. act as tumor suppressors and play a critical role in the repair of double-stranded DNA breaks.
- 61. The lifetime risk of ovarian cancer is approximately 20% to 40% for BRCA1 mutations, and 10% to 20% for BRCA2 mutation carriers. The majority of BRCA1-associated cancers are serous adenocarcinomas, with an average age at diagnosis of 48 years, whereas the mean age for BRCA2-associated ovarian cancers is 60 years. BRCA-associated cancers may have a more favorable course than sporadic ovarian cancer - improved response to platinum-based chemotherapy compared to women with sporadic disease
- 62. The hereditary nonpolyposis colorectal cancer (HNPCC) syndrome accounts for approximately 5% to 10% of all hereditary ovarian cancer cases. It is an autosomal dominant genetic syndrome characterized by - nonpolyposis colon cancer, - endometrial, - ovarian, - hepatobiliary, - upper gastrointestinal, and - genitourinary cancers. The risk of endometrial cancer 40% to 60% by age 70, compared to 1.5% in the general population. 3.5-fold increase in the risk of ovarian cancer in members of these families.
- 63. A germline mutation in one of five genes involved in DNA mismatch repair is responsible for the HNPCC phenotype: hMSH2 (chromosome arm 2p), hMLH1 (chromosome arm 3p), hPMS1 (chromosome arm 2q), hPMS2 (chromosome arm 7p), and hMSH6 (chromosome arm 2p). The majority of affected patients are found to have defects in either hMSH2 or hMLH1. The estimated cumulative risks for ovarian cancer by age 70 years can be as high as 24% for those with germline mutations in MLH1 and MSH2.
- 64. It has been suggested that chemoprophylaxis with oral contraceptives for 5 years might decrease ovarian cancer risk by 50% in both the general population and in high-risk women. For example, a case-controlled study of 207 known BRCA mutation carriers and their sister controls found a 60% reduction of ovarian cancer risk with oral contraceptive use. Other risk-reducing strategies such as tubal ligation and hysterectomy have also been associated with a reduced incidence of ovarian cancer among high-risk women.
- 65. Nonetheless, RRSO is currently the most effective preventative strategy to reduce ovarian cancer risk in patients with BRCA mutations. One study of 259 patients who underwent RRSO found a 96% decrease in ovarian cancers and an approximately 50% reduction in subsequent breast cancer compared to age-matched controls. However, there is a residual risk for primary peritoneal cancer in these high-risk women after prophylactic salpingo-oophorectomy.
- 66. STAGING Exploratory laparotomy serves three main purposes in the management of patients with suspected ovarian cancer. - laparotomy permits histologic confirmation of disease, - the extent of disease (staging), which is critical in determining whether postoperative treatment will be necessary, as well as to assess prognosis. - to permit debulking of as much tumor as possible, since patients who are optimally cytoreduced (defined as having less than or equal to 1-cm diameter residual tumor) have a better prognosis compared to those with greater amounts of residual disease. The staging system for ovarian cancer is defined by FIGO (updated in 2014) and is based on the findings at exploratory laparotomy .
- 67. FIGO TNM I T1 Tumor confined to ovaries or fallopian tube(s) IA T1a Tumor limited to 1 ovary (capsule intact) or fallopian tube; no tumor on ovarian or fallopian tube surface; no malignant cells in the ascites or peritoneal washings IB T1b Tumor limited to both ovaries (capsules intact) or fallopian tubes; no tumor on ovarian or fallopian tube surface; no malignant cells in the ascites or peritoneal washings IC Tumor limited to 1 or both ovaries or fallopian tubes, with any of the following: IC1 T1c1 Surgical spill IC2 T1c2 Capsule ruptured before surgery or tumor on ovarian or fallopian tube surface IC3 T1c3 Malignant cells in the ascites or peritoneal washings
- 68. II T2 Tumor involves 1 or both ovaries or fallopian tubes with pelvic extension (below pelvic brim) or peritoneal cancer IIA T2a Extension and/or implants on uterus and/or fallopian tubes IIB T2b Extension to other pelvic organs (bladder,rectum,pelvic side wall)
- 69. III T1/T2-N1 Tumor involves 1 or both ovaries or fallopian tubes, or primary peritoneal cancer, with cytologically or histologically confirmed spread to the peritoneum outside the pelvis and/ or metastasis to the retroperitoneal lymph nodes IIIA1 Positive retroperitoneal lymph nodes only (cytologically or histologically proven): IIIA1(i) Metastasis up to 10 mm in greatest dimension IIIA1(ii) Metastasis more than 10 mm in greatest dimension IIIA2 T3a2-N0/N1 Microscopic extrapelvic (above the pelvic brim) peritoneal involvement with or without positive retroperitoneal lymph nodes IIIB T3b-N0/N1 Macroscopic peritoneal metastasis beyond the pelvis up to 2 cm in greatest dimension, with or without metastasis to the retroperitoneal lymph nodes IIIC T3c-N0/N1 Macroscopic peritoneal metastasis beyond the pelvis more than 2 cm in greatest dimension, with or without metastasis to the retroperitoneal lymph nodes (includes extension of tumor to capsule of liver and spleen without parenchymal involvement of either organ)
- 70. IV Any T, any N, M1 Distant metastasis exluding peritoneal metastases IVA Pleural effusion with positive cytology IVB Parenchymal metastases and metastases to extra-abdominal organs (including inguinal lymph nodes and lymph nodes outside of the abdominal cavity)
- 71. BORDERLINE TUMORS Ovarian borderline tumors are epithelial neoplasms that are histologically distinguished from ovarian carcinomas by the absence of stromal invasion. also referred to as tumors of LMP, which reflects their indolent natural history. Although borderline tumors generally do not exhibit stromal invasion within the ovary, cells from the primary tumor mass can be shed into the peritoneal cavity and eventually form serosal implants that involve the bowel, omentum, and upper abdomen.
- 72. Serous borderline tumors are more common and may be bilateral in 10% to 20% of cases. Mucinous borderline tumors tend to be larger than their serous counterparts, are infrequently bilateral, and are occasionally associated with pseudomyxoma peritonei. Borderline malignant potential tumors are distinguished from benign cystadenomas by the presence of epithelial budding, multilayering of the epithelium, increased mitotic activity, and nuclear atypia. They are distinguished from epithelial carcinomas by the absence of stromal invasion.
- 73. approximately 20% of ovarian tumors diagnosed as borderline on frozen-section analysis prove to be carcinomas on review of the permanent section. Greater than 90% of patients with early stage borderline tumors are alive at 10 years, and >50% of patients with advanced disease experience long- term survival. The fact that survival does not appear to be improved by postoperative adjuvant treatment.
- 74. GERM CELL TUMORS OF THE OVARY less common than epithelial ovarian neoplasms, accounting for 2% to 3% of all ovarian cancers in Western countries. usually occurring in younger women, with a peak incidence in women in their early 20s. An increased Incidence found in Asian and black populations.(15%) divided into dysgerminoma, which is the female counterpart of male seminoma, and nondysgerminoma.
- 75. Clinical features: Abdominal pain, distention, pelvic fullness, and urinary symptoms are common in patients with germ cell tumors of the ovary. In a minority of patients, abdominal pain can be severe, usually the result of hemorrhage, rupture, or ovarian torsion. rapid growth of most ovarian germ cell tumors causes pain due to stretching of the ovarian capsule, often prompting the patient to seek medical attention while the tumor is still confined to the ovary.
- 76. In contrast to epithelial tumors, Stage I - 60% to 70%. Stages II and IV tumors are relatively uncommon, and Stage III - 25% to 30% Dysgerminoma and mature cystic teratoma may be bilateral in 10% to 15% of cases. More advanced disease may involve retroperitoneal lymph nodes and multiple peritoneal surfaces, although ascites is infrequent. Hematogenous spread to the liver, lung, and brain can be observed, especially with choriocarcinoma.
- 77. Serum levels of α-fetoprotein (AFP) and β–human chorionic gonadotropin (HCG) are often helpful. - endodermal sinus tumor (AFP elevation only), - embryonal carcinoma (both AFP and HCG elevation), or - choriocarcinoma (HCG elevation only). - pure immature teratoma of the ovary typically have normal levels of AFP and HCG, although the AFP may be elevated in 30% of cases.
- 78. choriocarcinoma have extreme elevation of the β- HCG that results in hyperthyroidism due to homology between β-HCG and thyroid-stimulating hormone. mature cystic teratoma may also present with hyperthyroidism related to tumor-derived secretion of thyroxine, produced by mature thyroid tissue present within the tumor itself (struma ovarii).
- 79. SEX CORD–STROMAL TUMORS Approximately 5% of all ovarian cancers. Stage I disease, which has an excellent prognosis. Granulosa cell tumors are the most common type of sex cord–stromal malignancy. These tumors may produce estrogenic or, less commonly, androgenic steroids. Granulosa cell tumors may secrete other factors such as Inhibin and Müllerian inhibitory substance, which can sometimes be useful as tumor markers.
- 80. The hormonal manifestations of sex cord–stromal neoplasms such as granulosa cell tumors vary depending on the age of the patient. Thus, granulosa cell tumors occurring in premenarchal girls may present with precocious puberty. whereas women in the reproductive years may present with amenorrhea or abnormal menses or may occasionally develop intra-abdominal hemorrhage that mimics an ectopic pregnancy. Postmenopausal women - with postmenopausal bleeding due to endometrial hyperplasia (or a separate uterine carcinoma), resulting from tumor-derived estrogen. Sertoli-Leydig cell tumors may present with symptoms of virilization.
- 81. PROGNOSTIC FACTORS FOR EPITHELIAL OVARIAN CANCER Clinicopathologic findings of prognostic value include -FIGO stage, -volume of residual disease after cytoreductive surgery, -histologic subtype (mucinous or clear cell histologic have a worse prognosis) -histologic grade, -age, and -malignant ascites.
- 82. Preoperative serum CA 125 levels frequently reflect the volume of disease and do not appear to have an independent effect on survival. However, postoperative CA 125 levels, both during and after completion of first-line chemotherapy, have prognostic value. Some investigators have demonstrated that normalization of the serum CA 125 levels after three cycles of chemotherapy is associated with more favorable outcome.
- 83. Thank you







![INTRODUCTION
Ovarian neoplasms consist of several histopathologic
entities; treatment depends on the specific tumor type.
Epithelial ovarian cancer comprises the majority of
malignant ovarian neoplasms (about 90%);
less common histopathologies, including
- ovarian low malignant potential [LMP] tumor,
- malignant germ cell neoplasms,
- carcinosarcomas (malignant mixed Müllerian
tumors [MMMTs] and
- malignant sex cord- stromal tumors.](https://image.slidesharecdn.com/caovarystagingetiologypathogenesis-171212163256/85/Ca-ovary-staging-etiology-pathogenesis-7-320.jpg)















































![ An ongoing trial is assessing screening for ovarian
cancer (UK Collaborative Trial of Ovarian Cancer
Screening [UKCTOCS]) using multimodality
screening with ultrasound and CA-125 versus either
ultrasound alone or no screening.
Preliminary results suggest that multimodality
screening is more effective at detecting early-stage
cancer.](https://image.slidesharecdn.com/caovarystagingetiologypathogenesis-171212163256/85/Ca-ovary-staging-etiology-pathogenesis-56-320.jpg)


























