Lec,7-plasma proteins
- 2. •PLASMA PROTEINS •BY •DR SHAMIM AKRAM •AP BIOCHEM AIMC
- 3. Biochemistry For Medics 7/11/2012 3
- 4. Plasma consists of water, electrolytes, metabolites, nutrients, proteins, and hormones. The concentration of total protein in human plasma is approximately 6.0–8.0 g/dL and comprises the major part of the solids of the plasma. The proteins of the plasma are a complex mixture that includes not only simple proteins but also conjugated proteins such as glycoproteins and various types of lipoproteins. Biochemistry For Medics 7/11/2012 4
- 5. Biochemistry For Medics 7/11/2012 5
- 6. Salting-out methods-three major groups— fibrinogen, albumin, and globulins—by the use of varying concentrations of sodium or ammonium sulfate. Electrophoresis- five major fractions Albumin α1 and α2 globulins β globulins γ globulins Biochemistry For Medics 7/11/2012 6
- 7. Biochemistry For Medics 7/11/2012 7
- 8. DENSITOMETER SCANNING (Estimate conc. of each fraction) • MM
- 10. PLASMA PROTEINS
- 12. Albumin (69 kDa) is the major protein of human plasma (3.4–4.7 g/dL) Makes up approximately 60% of the total plasma protein. About 40% of albumin is present in the plasma, and the other 60% is present in the extracellular space. Half life of albumin is about 20 days. Migrates fastest in electrophoresis at alkaline pH. precipitates last at full saturation in salting out methods(bcz of its small size and larger surface area). Biochemistry For Medics 7/11/2012 12
- 13. The liver produces about 12 g of albumin per day, representing about 25% of total hepatic protein synthesis and half its secreted protein. Albumin is initially synthesized as a preproprotein Its signal peptide is removed as it passes into the cisternae of the rough endoplasmic reticulum, a hexapeptide at the resulting amino terminal is subsequently cleaved off farther along the secretory pathway. Biochemistry For Medics 7/11/2012 13
- 14. Mature human albumin consists of one polypeptide chain of 585 amino acids and contains 17 disulfide bonds It has an ellipsoidal shape, which means that it does not increase the viscosity of the plasma as much as an elongated molecule such as fibrinogen does. Has a relatively low molecular mass about 69 kDa Has an iso-electric pH of 4.7 Biochemistry For Medics 7/11/2012 14
- 15. 1-Colloidal osmotic Pressure-albumin is responsible for 75–80% of the osmotic pressure of human plasma due to its low molecular weight and large concentration It plays a predominant role in maintaining blood volume and body fluid distribution. Hypoalbuminemia leads to retention of fluid in the tissue spaces(Edema) Biochemistry For Medics 7/11/2012 15
- 16. Edema Biochemistry For Medics 7/11/2012 16 Normal level- 3.5-5 G/dl Hypoalbuminemia-(<2g/dl) Results in fluid retention in the tissue spaces. Lowered level is seen in the following conditions- Cirrhosis of liver Malnutrition Nephrotic syndrome Burns Malabsorption Analbuminemia- congenital disorder Hyperalbuminemia- In conditions of fluid depletion (Haemoconcentration))
- 17. A/G RATIO
- 18. 2-Transport function- Albumin has an ability to bind various ligands, thus acts as a transporter for various molecules. These include- Biochemistry For Medics 7/11/2012 18 free fatty acids (FFA), calcium, certain steroid hormones, bilirubin, copper A variety of drugs, including sulfonamides, penicillin G, dicoumarol, phenytoin and aspirin, are also bound to albumin
- 19. 3-Nutritive Function Albumin serves as a source of amino acids for tissue protein synthesis to a limited extent, particularly in nutritional deprivation of amino acids. 4-Buffering Function-Among the plasma proteins, albumin has the maximum buffering capacity due to its high concentration and the presence of large number of histidine residues, which contribute maximally towards maintenance of acid base balance. 5-Viscosity- Exerts low viscosity Biochemistry For Medics 7/11/2012 19
- 20. Blood brain barrier- Albumin- free fatty acid complex can not cross the blood brain barrier, hence fatty acids can not be utilized by the brain. Loosely bound bilirubin to albumin can be easily replaced by drugs like aspirin/ceftriaxone/claferon. In new born if such drugs are given, the released bilirubin gets deposited in brain causing Kernicterus. Biochemistry For Medics 7/11/2012 20
- 21. Protein bound calcium Biochemistry For Medics 7/11/2012 21 Calcium level is lowered in conditions of Hypo- Albuminemia Serum total calcium may be decreased Ionic calcium remains same Tetany does not occur Calcium is lowered by 0.8 mg/dl for a fall of 1g/dl of albumin
- 22. Drug interactions— Two drugs having same affinity for albumin when administered together, can compete for available binding sites with consequent displacement of other drug, resulting in clinically significant drug interactions. Example-Phenytoin(anticonvulsant-used in epilepsy) dicoumarol(anticoagulant) interactions. Biochemistry For Medics 7/11/2012 22
- 23. Clinical Significance of Albumin • Albumin bind certain compounds ,prevent them crossing blood-brain barrier e.g.free fatty acids,bilirubin. • Albuminuria is excretion of albumin in urine e.g.nephrotic syndrome. • Microalbuminurea is excretion of albumin 30- 300mg/day in urine,clinically imp. Is for predition of future risk of renal disease. • Albumin is therapeutically used for burns & renal failure.
- 24. Globulins are separated by half saturation with ammonium sulphate Molecular weight ranges from 90,000 to 13,00,000 By electrophoresis globulins can be separated in to – α1-globulins α2-globulins β-globulins Y-globulins Biochemistry For Medics 7/11/2012 24
- 25. α and β globulins are synthesized in the liver. Y globulins are synthesized in plasma cells and B-cells of lymphoid tissues (Reticulo- endothelial system) Synthesis of Y globulins is increased in chronic infections, chronic liver diseases, auto immune diseases, leukemias, lymphomas and various other malignancies. Biochemistry For Medics 7/11/2012 25
- 26. They are glycoproteins Based on electrophoretic mobility , they are sub classified in to α1 and α2 globulins α1 globulins Examples- α1antitrypsin Orosomucoid (α1 acid glycoprotein binds progestron) α1-fetoprotein (AFP) Biochemistry For Medics 7/11/2012 26
- 27. α1-antitrypsin Biochemistry For Medics 7/11/2012 27 Also called α1-antiprotease It is a single-chain protein of 394 amino acids, contains three oligosaccharide chains It is the major component (> 90%) of the α 1 fraction of human plasma. It is synthesized by hepatocytes and macrophages and is the principal serine protease inhibitor of human plasma. It inhibits trypsin, elastase, and certain other proteases by forming complexes with them.
- 28. At least 75 polymorphic forms occur, many of which can be separated by electrophoresis The major genotype is MM, and its phenotypic product is PiM A deficiency of this protein has a role in certain cases (approximately 5%) of emphysema. This occurs mainly in subjects with the ZZ genotype, who synthesize PiZ, and also in PiSZ heterozygotes, both of whom secrete considerably less protein than PiMM individuals. Biochemistry For Medics 7/11/2012 28
- 29. Emphysema- Normally antitrypsin protects the lung tissue from proteases(active elastase) released from macrophages/bacteria Forms a complex with protease and inactivates it. In its deficiency, the active elastase destroys the lung tissue by proteolysis. Biochemistry For Medics 7/11/2012 29
- 30. Smoking and Emphysema-A methionine (residue 358) of α1-antitrypsin is involved in its binding to proteases. Smoking oxidizes this methionine to methionine sulfoxide and thus inactivates it. Affected molecules of α1-antitrypsin no longer neutralize proteases. This is particularly devastating in patients (eg, PiZZ phenotype) who already have low levels of α1-antitrypsin. The further diminution in α 1-antitrypsin brought about by smoking results in increased proteolytic destruction of lung tissue, accelerating the development of emphysema. Biochemistry For Medics 7/11/2012 30
- 31. Biochemistry For Medics 7/11/2012 31
- 32. Cirrhosis of Liver- In this condition, molecules of the ZZ phenotype accumulate and aggregate in the cisternae of the endoplasmic reticulum of hepatocytes. Aggregation is due to formation of polymers of mutant α 1-antitrypsin, the polymers forming via a strong interaction between a specific loop in one molecule and a prominent -pleated sheet in another (loop-sheet polymerization). By mechanisms that are not understood, hepatitis results with consequent cirrhosis (accumulation of massive amounts of collagen, resulting in fibrosis). Biochemistry For Medics 7/11/2012 32
- 33. Biochemistry For Medics 7/11/2012 33
- 34. Concentration in plasma- 0.6 to 1.4 G/dl Carbohydrate content 41% Marker of acute inflammation Acts as a transporter of progesterone Transports carbohydrates to the site of tissue injury Concentration increases in inflammatory diseases, cirrhosis of liver and in malignant conditions Concentration decreases in liver diseases, malnutrition and in nephrotic syndrome Biochemistry For Medics 7/11/2012 34
- 35. Present in high concentration in fetal blood during mid pregnancy Normal concentration in healthy adult- < 1µg/100ml Level increases during pregnancy Clinically considered a tumor marker for the diagnosis of hepatocellular carcinoma or teratoblastomas. Biochemistry For Medics 7/11/2012 35
- 36. Clinically important α2-globulins are- Haptoglobin Ceruloplasmin α2- macroglobulins Biochemistry For Medics 7/11/2012 36
- 37. It is a plasma glycoprotein that binds extracorpuscular hemoglobin (Hb) in a tight noncovalent complex (Hb-Hp). The amount of Haptoglobin in human plasma ranges from 40 mg to 180 mg of hemoglobin-binding capacity per deciliter. The function of Hp is to prevent loss of free hemoglobin into the kidney. This conserves the valuable iron present in hemoglobin, which would otherwise be lost to the body. Biochemistry For Medics 7/11/2012 37
- 38. The molecular mass of hemoglobin is approximately 65 kDa Hb-Hp complex has a molecular mass of approximately 155 kDa. Free hemoglobin passes through the glomerulus of the kidney, enters the tubules, and tends to precipitate therein (as can happen after a massive incompatible blood transfusion, when the capacity of haptoglobin to bind hemoglobin is grossly exceeded). However, the Hb-Hp complex is too large to pass through the glomerulus. Thus Hp helps to conserve iron. Biochemistry For Medics 7/11/2012 38
- 39. Concentration rises in inflammatory conditions Concentration decreases hemolytic anemias Half-life of haptoglobin is approximately 5 days, the half-life of the Hb-Hp complex is about 90 minutes, the complex being rapidly removed from plasma by hepatocytes. Thus, when haptoglobin is bound to hemoglobin, it is cleared from the plasma about 80 times faster than normally. The level of haptoglobin falls rapidly in hemolytic anemias. Free Hp level or Hp binding capacity depicts the degree of intravascular hemolysis. Biochemistry For Medics 7/11/2012 39
- 40. Copper containing α2-globulin Glycoprotein with enzyme activities It has a blue color because of its high copper content Carries 90% of the copper present in Plasma(rest of 10% carried by albumin). Each molecule of ceruloplasmin binds six atoms of copper very tightly, so that the copper is not readily exchangeable. Biochemistry For Medics 7/11/2012 40
- 41. Normal plasma conc 30mg/dL Enzyme activities are Ferroxidase, copper oxidase and Histaminase. Synthesized in liver in the form of apo ceruloplasmin, when copper atoms get attached it becomes Ceruloplasmin. Although carries 90% of the copper present in plasma. but it binds copper very tightly, so that the copper is not readily exchangeable. Albumin carries the other 10% of the plasma copper but binds the metal less tightly than does ceruloplasmin. Albumin thus donates its copper to tissues more readily than ceruloplasmin and appears to be more important than ceruloplasmin in copper transport in the human body. Biochemistry For Medics 7/11/2012 41
- 42. Normal level- 25-50 mg/dl Biochemistry For Medics 7/11/2012 42 Low levels of ceruloplasmin are found in Wilson disease (hepatolenticular degeneration),Menkes disease, due to abnormal metabolism of copper. The amount of ceruloplasmin in plasma is also decreased in liver diseases, mal nutrition and nephrotic syndrome.
- 43. Major component of α2 proteins Comprises 8–10% of the total plasma protein in humans. Tetrameric protein with molecular weight of 725,000. Synthesized by hepatocytes and macrophages Inactivates all the proteases and thus is an important in vivo anticoagulant. Carrier of many growth factors Normal serum level-130-300 mg/dl Concentration is markedly increased in nephrotic syndrome, since other proteins are lost through urine in this condition. Biochemistry For Medics 7/11/2012 43
- 44. β Globulins of clinical importance are – Biochemistry For Medics 7/11/2012 44 Transferrin C-reactive protein Haemopexin Complement C1q β Lipoprotein(LDL)
- 45. Transferrin (Tf) is a β 1-globulin with a molecular mass of approximately 76 kDa. It is a glycoprotein and is synthesized in the liver. About 20 polymorphic forms of transferrin have been found. It plays a central role in the body's metabolism of iron because it transports iron (2 mol of Fe3+ per mole of Tf) in the circulation to sites where iron is required, eg, from the gut to the bone marrow and other organs. Approximately 200 billion red blood cells (about 20 mL) are catabolized per day, releasing about 25 mg of iron into the body—most of which is transported by transferrin. Biochemistry For Medics 7/11/2012 45
- 46. There are receptors (TfR1 and TfR2) on the surfaces of many cells for transferrin. It binds to these receptors and is internalized by receptor-mediated endocytosis. The acid pH inside the lysosome causes the iron to dissociate from the protein. The dissociated iron leaves the endosome via DMT1 to enter the cytoplasm. ApoTf is not degraded within the lysosome. Instead, it remains associated with its receptor, returns to the plasma membrane, dissociates from its receptor, reenters the plasma, picks up more iron, and again delivers the iron to needy cells. Normally, the iron bound to Tf turns over 10–20 times a day. Biochemistry For Medics 7/11/2012 46
- 47. Biochemistry For Medics 7/11/2012 47
- 48. The concentration of transferrin in plasma is approximately 300 mg/dL. This amount of transferrin can bind 300 g of iron per deciliter, so that this represents the total iron-binding capacity of plasma. However, the protein is normally only one-third saturated with iron. In iron deficiency anemia, the protein is even less saturated with iron, whereas in conditions of storage of excess iron in the body (eg, hemochromatosis) the saturation with iron is much greater than one-third. Biochemistry For Medics 7/11/2012 48
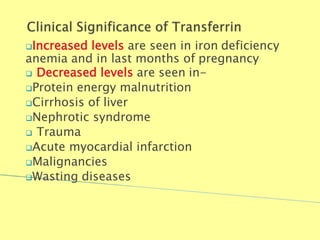
- 49. Increased levels are seen in iron deficiency anemia and in last months of pregnancy Decreased levels are seen in- Protein energy malnutrition Cirrhosis of liver Nephrotic syndrome Trauma Acute myocardial infarction Malignancies Wasting diseases Biochemistry For Medics 7/11/2012 49
- 50. So named because it reacts with C- polysaccharide of capsule of pneumococci Molecular weight of 115-140 kD Synthesized in liver Can stimulate complement activity and macrophages Acute phase protein- Concentration rises in inflammatory conditions Clinically important marker to predict the risk of coronary heart disease Biochemistry For Medics 7/11/2012 50
- 51. Molecular weight 57,000-80,000 Normal level in adults-0.5 to 1.0 gm/L Low level at birth, reaches adult value within first year of life Synthesized in liver Function is to bind haem formed from breakdown of Hb and other haemoproteins Low level- found in hemolytic disorders, at birth and drug induced High level- pregnancy, diabetes mellitus, malignancies and Duchenne muscular dystrophy Biochemistry For Medics 7/11/2012 51
- 52. First complement factor to bind antibody Binding takes place at the Fc region of IgG or Ig M Binding triggers the classical complement pathway Thermo labile, destroyed by heating Normal level – 0.15 gm/L Molecular weight-400,000 Can bind heparin and bivalent ions Decreased level is used as an indicator of circulating Ag –Ab complex. High levels are found in chronic infections Biochemistry For Medics 7/11/2012 52
- 53. They are immunoglobulins with antibody activity They occupy the gamma region on electrophoresis Immunoglobulins play a key role in the defense mechanisms of the body There are five types of immunoglobulins IgG, IgA, IgM, IgD, and IgE. Biochemistry For Medics 7/11/2012 53
- 55. Immunoglobulin Major Functions IgG IgA IgM IgD IgE Main antibody in the secondary response. Phagocytosis of bacteria, Fixes complement, neutralizes bacterial toxins and viruses and crosses the placenta. Secretory IgA prevents attachment of bacteria and viruses to mucous membranes. Does not fix complement. Produced in the primary response to an antigen. Fixes complement. Does not cross the placenta. Antigen receptor on the surface of B cells. Uncertain. Found on the surface of many B cells as well as in serum. Mediates immediate hypersensitivity DefeBniodchsemaistgryaFionrMsetdicws o7r/m11/2i0n1f2ections. Doe47s
- 56. Also called clotting factor1 Constitutes 4-6% of total protein Precipitated with 1/5 th saturation with ammonium sulphate Large asymmetric molecule Highly elongated with axial ratio of 20:1 Imparts maximum viscosity to blood Synthesized in liver Made up of 6 polypeptide chains Chains are linked together by S-S linkages Amino terminal end is highly negative due to the presence of glutamic acid Negative charge contributes to its solubility in plasma and prevents aggregation due to electrostatic repulsions between the fibrinogen molecules. Biochemistry For Medics 7/11/2012 56
- 57. Name Compounds transported Albumin Fatty acids, bilirubin, hormones, calcium, heavy metals, drugs etc. Prealbumin-(Transthyretin) Steroid hormones thyroxin, Retinol Retinol binding protein Retinol (Vitamin A) Thyroxin binding protein(TBG) Thyroxin Transcortin(Cortisol binding protein) Cortisol and corticosteroids Haptoglobin Hemoglobin Hemopexin Free haem Transferrin Iron HDL(High density lipoprotein) Cholesterol (Tissues to liver) LDL(Low density lipoprotein) Cholesterol(Liver to tissues) Biochemistry For Medics 7/11/2012 57
- 58. The levels of certain proteins may increase in blood in response to injury,inflammation,infection and neoplastic conditions, these are called Acute phase proteins. Examples- Biochemistry For Medics 7/11/2012 58 C- reactive proteins Ceruloplasmin Alpha -1 antitrypsin Alpha 2 macroglobulins Alpha-1 acid glycoprotein
- 59. The levels of certain proteins are decreased in blood in response to certain inflammatory processes. Examples- Biochemistry For Medics 7/11/2012 59 Albumin Transthyretin Retinol binding protein Transferrin
- 60. 1) Bence – Jone’s proteins Biochemistry For Medics 7/11/2012 60 Abnormal proteins- monoclonal light chains Present in the urine of a patient suffering from multiple myeloma (50% of patients) Molecular weight 45,000 Identified by heat coagulation test-ppt at 50- 60C,dissolve on further raising temp.reappear when cooled down to 60 C Best detected by zone electrophoresis and immunoelectrophoresis 2)Cryoglobulins These proteins coagulate when serum is cooled to very low temperature Commonly monoclonal IgG or IgM or both Increased in rheumatoid arthritis, multiple myeloma, lymphocytic leukemia, lymphosarcoma and systemic lupus erythematosus
- 61. Nutritive Fluid exchange Buffering Binding and transport Enzymes Hormones Blood coagulation Viscosity Defense Reserve proteins Tumor markers Antiproteases Biochemistry For Medics 7/11/2012 61
- 62. Hyperproteinemia- Levels higher than 8.0gm/dl Causes- Biochemistry For Medics 7/11/2012 62 Hemoconcentration- due to dehydration, albumin and globulin both are increased Albumin to Globulin ratio remains same. Causes- Excessive vomiting Diarrhea Diabetes Insipidus Pyloric stenosis or obstruction Diuresis Intestinal obstruction
- 63. 1)Polyclonal - Biochemistry For Medics 7/11/2012 63 Chronic infections Chronic liver diseases Sarcoidosis Auto immune diseases 2) Monoclonal Multiple myeloma Macroglobulinaemia Lymphosarcoma Leukemia Hodgkin’s disease
- 64. Decease in total protein concentration Hemodilution- Both Albumin and globulins are decreased, A:G ratio remains same, as in water intoxication Hypoalbuminemia- low level of Albumin in plasma • Causes- Nephrotic syndrome Protein losing enteropathy Severe liver diseases Mal nutrition or malabsorption Extensive skin burns Pregnancy Malignancy Biochemistry For Medics 7/11/2012 64
- 65. Losses from body- same as albumin- through urine, GIT or skin Decreased synthesis Transient neonatal Primary genetic deficiency Secondary – drug induced (Corticosteroid therapy), uremia, hematological disorders AIDS(Acquired Immuno deficiency syndrome) Biochemistry For Medics 7/11/2012 65