MONOCHORIONIC TWIN PREGNANCY detailed.pptx
- 2. Timeline • Introdution • Background • Zygosity & Chorionicity • Diagnosis • Complications of Twin • Complications specific to Monochorionic Twin • Management • Bottomline
- 3. Introduction • A monochorionic pregnancy is a multiple pregnancy, most commonly a twin pregnancy (99% of cases), in which babies are dependent on a single shared placenta • Placental anastomoses conjoining the fetal circulations • Monochorionic placentation can also occur in rarer, higher-order multiples, especially triplets
- 4. Introduction contd... • Increase in all types of multiple pregnancies with: the use of assisted reproductive technology to defer pregnancy to a later maternal age • Day 5 blastocyst transfers seems to be associated with a significantly higher rate of monozygotic twinning compared with cleavage stage day 3 transfers
- 5. What is twin pregnancy • Two or more fertilization event • Single fertilization followed by splitting of zygote • Combination of both
- 6. Background Incidence of twin: 1 in 52 or 1.9% Incidence is highest in Nigeria (1 in 20) and lowest in Japan (1 in200) Frequency of monozygotic twin births: 1 per 250 births (relatively constant worldwide) In India:1 in 80 Hellin’s rule: Twin 1 in 80 Triplet 1 in 802 Quadruplets 1 in 803 & so on
- 7. Genesis of Monozygotic twins
- 8. Genesis of Monozygotic twins (contd…) • Dizygotic: Always DCDA • Monozygotic: Outcome depends on the time of division Timing of cleavage Placenta’s membrane status Percentage of monozygotic twin <72hrs DCDA 25-30% 4-8 DAYS MCDA 70-75% 8-12 DAYS MCMA 1-2% >13DAYS SIAMESE/ CONJOINED <1%
- 9. Determination of Zygosity Placental examination • Dichorionic diamniotic placenta • 2 layers of amnions with intervening chorion • Monochorionic diamniotic placenta • Only 2 layers of amnions without intervening chorion • Monochorionic monoamniotic placenta • No intervening membrane
- 10. Determination of Zygosity (contd...) • Confirmatory test for zygosity is DNA fingerprinting If monochorionic diamniotic(MCDA) or monochorionic monoamniotic(MCMA) Monozygotic If dichorionic diamniotic (DCDA) ? → →
- 11. Determination of Chorionicity • At 11 +2 – 14+1 weeks of gestation (CRL 45–84 mm) sonographic assessment of chorionicity is determined using following four features: 1. Number of placental masses 2. The appearance of the intertwin membrane attachment to the placenta 3. Thickness of that membrane 4. Fetal gender
- 12. Determination of Chorionicity (contd…) • Two separate placentas → Dichorionic (3% of Monochorionic twin have Bilobed placenta ) Single placenta → may be Dichorionic (two fused placenta) or Monochorionic • Sex discordance indicates Dichorionicity • Thickness of dividing membrane : ≥2 mm → Dichorionic pregnancy < 2 mm → Monochorionic pregnancy
- 13. Determination of Chorionicity (contd...) • Dichorionic pregnancy → Membrane composed of four layers 2 amnions and 2 chorions • Monochorionic pregnancy → Membrane composed of 2 layers amnion and amnion • The twin peak sign or lambda sign → Dichorionic twin • T sign → Monochorionic twin
- 14. Determination of Chorionicity (contd…) Done on USG between 11 +2 – 14+1 weeks of gestation 1. Two separate developing placentas 2. Twin peak or Lamda sign (Dichorionic) T sign (Monochorionic) 3. Thickness of the intertwin membrane(<2mm -- Monochorionic)
- 15. Twin Peak Sign • It appears as a triangular projection of placental tissue extending from the main placental mass into the inter-twin membrane
- 16. T Sign • The "T sign" is characterized by a thin membrane intersecting the placenta at a right angle, resembling the letter "T"
- 17. Nomenclature of Twins • On ultrasound, the fetuses in twin pregnancies should be be assigned nomenclature • Fetus A should be with amniotic sac closest to the maternal cervix • Document cord insertions and relation to placenta • Labelling critical for anomaly tracking and delivery planning • Standardize labelling from early scans for clarity upper lower left right
- 18. Screening for Chromosomal Anomalies in Monochorionic Twin Pregnancies • Aneuploidy screening: Nuchal translucency + first-trimester serum markers (combined screening test) Done at 11 +2 – 14+1 weeks of gestation (CRL 45–84 mm) • Sensitivity - 90% • False-positive rate (up to 10%) is higher than in singletons (2.5%) and dichorionic twins (5%)
- 19. Screening for Chromosomal Anomalies in Monochorionic Twin Pregnancies (contd...) • Women who ‘miss’ or who have unsuccessful 1st trimester screening Second-trimester screening with quadruple test to be done DR: 80% and a false-positive rate of 3% • Cell free fetal (cff) DNA testing (also known as NIPT), contingent on the results of the first-trimester combined test for major autosomal trisomies also recommended
- 20. Increased Nuchal Translucency • Associated with TTTS, cardiac dysfunction, congenital defects • 20% discordance in NT values increases complication risk • NT >95th percentile: consider genetic testing and anomaly scan • Predictive of structural anomalies even without aneuploidy • Recommend combined screening test (NT + serum markers) • Early referral to fetal medicine when increased NT noted
- 21. Screening for Structural Abnormalities in Monochorionic Twin Pregnancies • Incidence of anomalies is higher in monozygotic twins due to the unusual nature of the cleavage of the conceptus • Monozygotic twins are monochorionic in 70% of cases; hence the higher rates of anomaly in monochorionic twins • Anomalies specific to monozygotic twins are often midline such as holoprosencephaly, neural tube defects and cardiac abnormalities
- 22. Screening for Structural Abnormalities in Monochorionic Twin Pregnancies (contd..) • All monochorionic twins should undergo a routine detailed ultrasound scan between 18 +0 and 20 +6 weeks of gestation, which includes extended views of the fetal heart anatomy
- 23. Optimum Ultrasound Regimen for Monochorionic Twin Pregnancies • Fetal ultrasound assessment should take place every 2 weeks in uncomplicated monochorionic pregnancies from 16 +0 weeks of gestation onwards until birth
- 24. The antenatal and ultrasound visit schedule for monochorionic twins
- 25. Parameters to be seen at each Ultrasound • Liquor volume in each of the amniotic sac • The deepest vertical pocket (DVP) depth • Umbilical artery pulsatility index(UA-PI) at every scan from 16 +0 weeks • Middle cerebral artery peak systolic velocity (MCA PSV) from 20 weeks • Fetal bladders should also be visualised • From 16 +0 weeks of gestation, fetal biometry, estimated fetal weight (EFW) and the difference in EFW (as there is risk of sGR)
- 26. USG between 16+0 to 26+0 weeks of gestation at 2 weekly intervals For detection of TTTS USG after 26+0 weeks Mainly focus on detection of sGR or concordant growth restriction USG after 20 weeks MCA PSV done To detect TAPS
- 27. Complications of Twin Pregnancy • Maternal Complications During pregnancy: Nausea and vomiting Anaemia Unexplained weight gain Preeclampsia (25%) Hydramnios (10%): severe hydramnios can lead to obstructive uropathy Antepartum haemorrhage
- 28. Complications of Twin Pregnancy (contd…) Malpresentation Preterm labour (50%) Mechanical distress Palpitation Dyspnea Varicosities & haemmorhoids
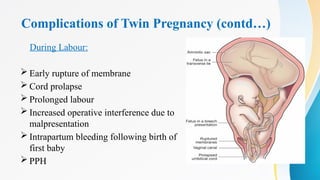
- 29. Complications of Twin Pregnancy (contd…) During Labour: Early rupture of membrane Cord prolapse Prolonged labour Increased operative interference due to malpresentation Intrapartum bleeding following birth of first baby PPH
- 30. Complications of Twin Pregnancy (contd…) During puerperium: Retained placenta Subinvolution Infection due to operative interference Lactation failure Depression
- 31. Complications of Twin Pregnancy (contd…) Fetal complications Spontaneous abortion Prematurity Discordant growth & Growth restriction Low birth weight Intrauterine fetal death Fetal anomalies: anencephaly, microcephaly, holoprosencephaly, neural tube defects, cardiac anomalies Asphyxia & stillbirth Long term: Cognitive delay, cerebral palsy
- 32. Complications specific to Monochorionic Twins • Twin twin transfusion syndrome (TTTS) • Twin anaemia polycythemia sequence (TAPS) • Selective growth restriction (sGR) • Twin reversed arterial perfusion (TRAP) sequence • Single fetal demise • Conjoined twin
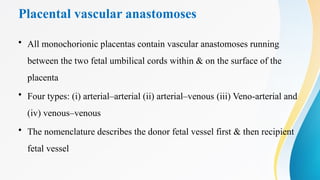
- 33. Placental vascular anastomoses • All monochorionic placentas contain vascular anastomoses running between the two fetal umbilical cords within & on the surface of the placenta • Four types: (i) arterial–arterial (ii) arterial–venous (iii) Veno-arterial and (iv) venous–venous • The nomenclature describes the donor fetal vessel first & then recipient fetal vessel
- 34. Placental vascular anastomoses (contd...) • Anastomosis are also 2 types: Superficial and Deep • Superficial anastomoses → bidirectional and balanced • Deep anastomoses → unidirectional and unbalanced • AA and VV anastomoses are superficial & end to end → allow bidirectional blood flow & do not cause TTTS • AV and VA anastomoses are deep → cause haemodynamic imbalance between the fetal circulations • AA anastomoses are protective
- 35. Twin to twin Transfusion Syndrome (TTTS) • Complicates up to 15%–20% of monochorionic twins (mostly MCDA twins) • Placenta has a predominance of unidirectional, arterial–venous anastomoses • Cause haemodynamic imbalance within the fetal circulations • Adversely affect fetal cardiac function, fetoplacental perfusion and causing fetal endocrine dysfunction • Severity is graded by Quintero staging
- 36. Pathophysiology of TTTS The three major factors in the pathogenesis of TTTS are: 1. Unbalanced flow volume 2. Release of vasoactive mediators 3. Lack of AA anastomosis
- 37. Vascular Anastomosis Superficial Anastomosis Deep Anastomosis • Most common • Most commonly: Artery-artery anastomosis • Bidirectional flow • No permanent donor/recipient No TTTS • Most commonly seen in MCMA Twin A Twin B • Most common type: Artery-Vein anastomosis • At the capillary level • Unidirectional flow Permanent Donor Permanent Recipient TTTS • Most commonly seen in MCDA Twin A Twin B Pathophysiology of TTTS (contd…)
- 38. Pathophysiology of TTTS (contd…) • AV anastomoses → chorionic plate surface arterial vessels from one twin and chorionic plate surface venous vessels from other twin connect in the underlying cotyledon • The cotyledon is supplied by the artery of one twin & blood is drained through vein of the co-twin • Lack of AA anastomoses aggravate the condition
- 39. Pathophysiology of TTTS (contd…) • Increased natriuretic peptides in recipient worsen effects • Imbalance activates RAAS in donor twin • Donor twin: hypovolemia → oliguria → oligohydramnios → stuck twin • Recipient twin: hypervolemia → stretching of cardiac atria→release of ANP→Stretching of ventricle → release of BNP→vasodilation & natriuresis → inhibition of RAAS → polyuria → polyhydramnios
- 40. Pathophysiology of TTTS (contd…) Classically, the donor twin is smaller, pale & growth restricted Recipient sibling is larger, polycythemic and has volume excess, may suffer hyperviscosity and occlusive complications, severe hyperbilirubinemia and kernicterus (due to polycythemia)
- 41. Pathophysiology of TTTS (contd…) Fetal brain damage: • Cerebral palsy, microcephaly, porencephaly, and multicystic encephalomalacia are serious complications Donor twin Anemia & hypotension→ischemia→cavitary brain lesion Recipient twin Blood pressure instability & episodes of profound hypotension→ischemia→brain damage
- 42. Pathophysiology of TTTS (contd…) Death of one twin in TTTS Severe hypotension in surviving twin Irreversible brain damage of surviving twin that makes successful intervention for the survivor nearly impossible
- 43. Placental Vascular Anastomoses in TTTS The following color code was applied for injection. Left twin: yellow = artery, blue = vein; right twin: red = artery, green = vein Part of the arterial network of the right twin is filled with yellow dye, due to the presence of a small artery-to- artery anastomosis (arrow) Shared placenta from pregnancy complicated by twin-twin transfusion syndrome
- 44. Quintero Staging of TTTS Stage I • A significant discordance in amniotic fluid volume • Oligohydramnios with DVP < 2 cm in donor sac • Polyhydramnios in the recipient sac (DVP > 8 cm before 20+0 weeks of gestation and > 10 cm after 20+0 weeks of gestation) • The Donor bladder is visible on ultrasound and the Umbilical Artery Doppler velocimetry is normal
- 45. Quintero Staging of TTTS (contd...) Stage II • The bladder is not visible on ultrasound • Severe oligohydramnios due to anuria of the donor twin • Umbilical Artery Doppler studies are minimally abnormal Stage III • Abnormal doppler studies in either the donor or recipient fetus (or both) • Abnormal (absent or reversed) UA doppler and/or abnormal venous doppler velocities in the recipient
- 46. Quintero Staging of TTTS (contd...) Stage IV • The presence of fetal effusions (ascites, pericardial or pleural effusions) with scalp oedema or overt hydrops fetalis present in either twin (but usually in the recipient) Stage V • One or both babies have died (not amenable to therapy)
- 47. Twin anaemia polycythemia sequence (TAPS) • Up to 2% of uncomplicated monochorionic diamniotic (MCDA) and ~13% of monochorionic twins post-fetoscopic laser ablation • Signs of fetal anaemia in the donor and polycythaemia in the recipient • Without significant oligohydramnios/polyhydramnios • Types of TAPS: Spontaneous TAPS Post laser ablation TAPS (develops within 5 weeks of the procedure)
- 48. Pathophysiology of TAPS Spontaneous TAPS: • Few minuscule arterio-venous vascular anastomoses • Unbalanced unidirectional slow transfusion of red blood cells through very small (<1 mm diameter) placental AV anastomoses • Large intertwin Hb difference • 5-15 ml of RBC transfusion in 24 hours from donor to recipient twin • Donor twin: Anaemia → hydrops fetalis • Recipient twin: Polycythemia → fetal and placental thrombosis
- 49. Pathophysiology of TAPS (contd...) • The slowness of the process allows haemodynamic compensation, hence there is absence of amniotic fluid discordance • Donor twin → increased MCA PSV • Recipient twin → decreased MCA PSV • The difference in MCA PSV ≥ 1.0 MoM
- 50. Pathophysiology of TAPS (contd...) Post laser ablation TAPS: • Sequelae of use of laser ablation for treatment of TTTS • Discordance of liquor volumes must be excluded, as if present would indicate a recurrence of TTTS (most often due to treatment failure)
- 51. Diagnosis of TAPS • 5 stages: I (discordant MCA-PSV) to V (IUFD) • Can be diagnosed antenatally or postnatally • Antenatal: MCA-PSV ≥ 1.5 MoM (donor) ≤ 0.8 MoM (recipient) or Difference in MCA PSV ≥ 1.0 MoM • Postnatal: Haematological diagnostic criteria- Inter-twin haemoglobin difference > than 80 g/L Reticulocyte ratio >1.7 Confirm with placental histopathology postnatally
- 52. Discordant Twin • Significant intrauterine fetal size discordance in monochorionic twins: Difference in estimated fetal weight [EFW] of greater than 20% and Smaller twin with EFW or abdominal circumference (AC) on ultrasound of < 10th centile for gestation • It is associated with marginally increased perinatal risk • Calculate percentage EFW discordance using the following formula: [larger twin EFW—smaller twin EFW]/larger twin EFW) × 100
- 53. Selective Growth Restriction (sGR) • When the selective fetal discordance is greater than 25%, it is termed ‘selective growth restriction’ (sGR) • Present in up to 20% of monochorionic twins 60% of monochorionic twins complicated by TTTS • Due to unequal placental sharing or perfusion • UA Doppler vital for classification • Associated with fetal demise and neurologic injury • Early recognition improves intervention planning
- 54. Diagnosis of Selective Growth Restriction (sGR) Estimated fetal weight [EFW] of one twin is less than 3rd centile OR there are 2 of the following 3 parameters— (i) A growth discordance of greater than 25% difference in EFW between the fetuses (ii) The smaller fetus having an EFW or AC less than 10th centile for gestation (iii) Abnormal umbilical artery (UA) Doppler of the smaller fetus (UA-PI of greater than 95th centile or shows absent or reversed end-diastolic velocity)
- 55. sGR Types Based on UA Doppler • Type I: Growth discordance but positive diastolic velocities in both fetal umbilical arteries • Type II: Growth discordance with absent or reversed end-diastolic velocities (AREDV) in one or both fetuses • Type III: Growth discordance with “cyclical” umbilical artery diastolic waveforms
- 56. Diagnostic difficulty in sGR • May be a diagnostic ‘overlap’ between mild TTTS and sGR • Liquor volumes as DVP should be measured to differentiate from TTTS Amniotic fluid (DVP) in TTTS Amniotic fluid (DVP) in sGR Donor twin → oligohydramnios Recipient twin → polyhydramnios Oligohydramnios in one of the amniotic sacs Normal liquor in the other amniotic sac
- 57. Prognosis of baby with sGR • Type I sGR: Relatively good, more than 90% perinatal survival • Type II sGR: High risk (up to 29%) of intrauterine demise of the growth-restricted twin and/or preterm birth • Type III sGR: Smaller twin: 10%–20% risk of the unexpected fetal demise (even if stable USG and/or normal CTG hours or days before) Larger twin: 10%–20% risk of neurological injury
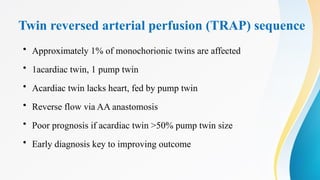
- 58. Twin reversed arterial perfusion (TRAP) sequence • Approximately 1% of monochorionic twins are affected • 1acardiac twin, 1 pump twin • Acardiac twin lacks heart, fed by pump twin • Reverse flow via AA anastomosis • Poor prognosis if acardiac twin >50% pump twin size • Early diagnosis key to improving outcome
- 59. Pathophysiology of TRAP Within the single shared placenta, arterial perfusion pressure of the donor twin exceeds that of the recipient twin The recipient receives reversed blood flow containing deoxygenated arterial blood from its co-twin “Used” arterial blood reaches the recipient twin through its umbilical arteries preferentially goes to its iliac vessels Only the lower body is perfused Disrupted growth and development of the upper body
- 60. Pathophysiology of TRAP (contd...) • Failed head growth → Acardius acephalus • Partially developed head with identifiable limbs → Acardius myelacephalus • Failure of any recognizable structure → Acardius amorphous Normal donor twin supports its own circulation & also must pump blood to the acardiac recipient Cardiomegaly and high-output heart failure in donor twin
- 62. TRAP Diagnosis • Ultrasound shows absent cardiac activity in acardiac twin • Reversed umbilical artery flow in acardiac twin • Large size discrepancy with pump twin • Risk stratification by acardiac:pump size ratio • Subcutaneous edema, abnormal morphology typical • Confirmed via Doppler and structural scan
- 63. Single Fetal Demise • IUFD in one twin affects co-twin via shared circulation • After a single fetal death in a monochorionic pregnancy: Risks of death to the surviving twin 15% Risk of neurological abnormality 26%
- 64. Fate of Surviving Twin after Single Fetal Demise One twin of an affected pregnancy dies Blood is transfused from high- pressure vessels of the living twin through anastomoses to low resistance vessels of the dead twin Hypovolemia & ischemic brain damage in the survivor Emboli of thromboplastic material originating from the dead fetus Neurological injury to the survivor Harm to the survivor
- 65. Single Fetal Demise (contd…) Diagnosing Neurological Damage: • MRI with diffusion-weighted imaging (DWI) • Best after 4 weeks post-IUFD • Detects ischemic lesions earlier than ultrasound • Ultrasound may miss early hypoxic injury • Types: porencephaly, encephalomalacia, hydrocephalus • Important for prognostication and parental counselling
- 66. Management of Twin Pregnancy
- 67. Antenatal Care Principles • Specialist-led care recommended • Regular emotional support and reassurance • Early counselling regarding twin risks • Discuss screening complexity and options at booking • Tertiary care referral for complications • Nutritional advice, weight monitoring & hydration
- 68. Supplements and Aspirin Use • Iron folic acid & calcium supplementation • Vitamin D supplementation if deficient • Aspirin 100–150 mg from 12–36 weeks as per guideline • Reduces risk of preeclampsia and placental dysfunction • Encourage compliance and monitor blood pressure regularly
- 69. Antenatal Visit Schedule • Determination of chorionicity & screening for chromosomal anomaly at 11 +2 – 14+1 weeks of gestation • Every 2-week scans from 16 to 36 weeks • Biometry, amniotic fluid (DVP), Doppler studies • Cervical length monitoring at 20–24 weeks • Detailed anomaly scan at 20 weeks • Delivery planning at 36 weeks if uncomplicated
- 70. Management of Specific Complications of Monochorionic Twin
- 71. Management of TTTS • Stage I: Expectant with close follow-up • Stages II–IV: Fetoscopic laser coagulation (preferred) Laser superior to amnioreduction for survival/neuroprotection Post-26 weeks: amnioreduction may be considered • Stage V: Palliative care, no active intervention • Decision guided by gestational age and expertise availability TTTS presenting before 26 +0 weeks of gestation should be treated by fetoscopic laser ablation rather than amnioreduction or septostomy. After 26 +0 weeks of gestation care should be individualised T
- 72. Foetoscopic Laser Types • Selective: Coagulates only visible AV connections • Sequential: Prioritizes AV over AA/VV anastomoses • Solomon: Equatorial laser dichorionisation Coagulate the entire vascular equator of the placenta, effectively creating a barrier between the twins' circulations and minimizing the risk of residual anastomoses
- 73. Solomon Technique Advantage 1. Improved survival rate 2. Reduces the risk of residual anastomoses→ decreased recurrence of TTTS or the development of TAPS Risk 1. Higher risk of placental damage 2. Placental abruption 3. PROM, PPROM, preterm labour
- 75. Post-laser Follow-up • Weekly ultrasound assessment (including examination of the fetal brain, heart and limbs) • Serial measurements of UA-PI, MCA PSV and Ductus Venosus Doppler velocities • After 2 weeks post-treatment, the ultrasound interval may be increased to every 2 weeks (UA-PI, MCA PSV, DVP and EFW) • In treated TTTS pregnancies, an ultrasound examination of the fetal heart should be performed by the fetal medicine specialist to exclude functional and acquired structural heart anomalies
- 76. Outcomes of Laser Therapy • 70–88% survival of at least one twin • Both twins survive in ~60% cases • Reduced rates of neurological damage compared to amnioreduction • TAPS can still occur after incomplete coagulation • Higher complications if done <17 or >26 weeks • Early intervention improves prognosis
- 77. Amnioreduction • Reduces intra-amniotic pressure • Improves uteroplacental perfusion transiently • Not curative—symptomatic relief only • Indicated post-26 weeks or where laser unavailable • Requires multiple procedures often • Risks include PPROM, infection, fetal loss
- 78. Septostomy • Intentional rupture of intertwin septum • Equalizes amniotic pressures between sacs • May create monoamniotic environment → cord entanglement • Rarely used due to complications • May reduce frequency of repeat interventions • Limited evidence for routine use
- 79. Timing of Delivery in Monochorionic Twin with TTTS • Delivery of monochorionic twin pregnancies with treated TTTS should be at 36 completed weeks of gestation, unless there are complications • The role of delayed umbilical cord clamping is controversial because of the theoretical risks of feto-fetal transfusion with ‘intact’ placental anastomoses
- 80. TAPS Management • Care options may include: (i) Conservative management with close observation and surveillance (ii) In utero transfusion [of the anaemic twin] with or without exchange transfusion [of the polycythaemic fetus] (iii) Fetoscopic laser ablation (iv) Selective termination of pregnancy (in some circumstances)
- 81. TAPS: Neonatal Outcomes • Common issues: anemia, polycythemia, thrombocytopenia • Donor twin: Transfusion needs postnatally • Recipient twin: Polycythemia & hyperviscosity: limb ischemia & NEC risk • Neurologic impairment in 10–17% survivors • No difference in impairment between donor/recipient • Close pediatric follow-up essential
- 82. Management of sGR • Evaluation in a fetal medicine centre • Ultrasound at an interval of 2 weeks with fetal Doppler assessment (UA PI, MCA PI, PSV and Ductus venosus) weekly • In early-onset sGR with poor fetal growth velocity and abnormal Doppler → selective reduction or fetoscopic laser ablation considered to save the appropriately grown co-twin
- 83. Management of sGR (contd...) • Longer ‘latency period’ between diagnosis and timing of birth in MC twins complicated by sGR because of: The placental anastomoses in monochorionic twins paradoxically may be beneficial for the smaller twin as a transfusion from the larger twin may compensate for the placental insufficiency Associated artery–artery anastomoses also helps Latency period is 10 weeks compared to 3-4 weeks in FGR
- 84. Management of sGR (contd...) Timing of birth: Following are considered: Abnormal DV Doppler waveforms Computerised CTG (STV) Fetal biometric measurement and growth velocities • In type I sGR → 34 +0 –35 +6 weeks of gestation (if satisfactory fetal growth velocity and normal UA doppler) • In type II and III sGR → 32 weeks of gestation (unless fetal growth velocity is significantly abnormal or there is a worsening of the fetal doppler)
- 85. Management of sGR (contd...) • Inform parents that in sGR and TTTS (even after successful treatment) there can be acute and serious complications like Sudden transfusional events (neither predictable nor preventable) Neurological morbidity • Despite regular monitoring, there may still be adverse perinatal outcomes
- 86. TRAP Management • Expectant management if small acardiac twin • Intervene if acardiac twin large or pump twin shows cardiovascular impairment • Cord occlusion or RFA between 12–16 weeks ideal • Goal: Prevent pump twin heart failure or IUFD • TRAPIST trial evaluating early vs late intervention • ~80% pump twin survival with intervention
- 87. Single Fetal Demise: Management • Weekly monitoring: CTG, MCA Doppler, BPP • Coagulation profile at 1 and 3 weeks post-death • Deliver at 34–36 weeks if stable • Timing and mode of birth should be individualised • Corticosteroids for lung maturity • Avoid invasive interventions unless indicated • Involve multidisciplinary team (FMU, neurology, neonatology)
- 88. Timing of Birth in Uncomplicated MC Twin • Plan delivery at 36+0 weeks if uncomplicated • Earlier if complications: TTTS, sGR, IUFD • If preterm birth planned : (i) up to 34 +6 weeks of gestation → Antenatal corticosteroid (ii) before 34 +0 weeks of gestation → Magnesium sulfate • Avoid delay beyond 36 weeks due to IUFD risk • Mode of delivery depends on fetal presentation and parity • Vaginal delivery unless specific indication for CS • Decision based on risks and parental preference
- 89. Complications Specific to Monoamniotic Twin • Cord entanglement: The umbilical cords of the twins becoming intertwined & restrict blood flow and oxygen supply to one or both fetuses • Diagnosed by colour doppler • Interlocking of twin: Chin of the aftercoming head of 1st twin, presented by breech, is prevented from coming into the pelvis by the 2nd twin presented by vertex (Rare: 1 in 800)
- 90. Timing of Delivery in MCMA Twin • MCMA twins have a high risk of fetal demise and should be a planned caesarean birth between 32 +0 and 34 +0 weeks of gestation
- 91. Delivery of Locked Twin Delivery of aftercoming head of first twin is done followed by pushing the head of second twin out of the pelvis. If fails CS is done & after unlocking 2nd twin is delivered abdominally & 1st twin vaginally By Zavanelli manoeuvre the first twin is returned to pelvis & CS is done to deliver both babies If 1st foetus is dead, decapitation of first twin and delivered vaginally, then delivery of second twin & finally delivery of the head of first twin is accomplished If fails If fails
- 92. Indication of Urgent Delivery of Second Twin • Severe vaginal bleeding • Cord prolapse • Inadvertent use of IV oxytocics with the delivery of anterior shoulder of the first baby • First baby delivered under general anaesthesia • Appearance of fetal distress
- 93. Bottomline • Incidence of twins is on the rise • Being a high risk condition, requires vigilant management. • Chorionicity determination is important • Complications are more in monochorionic twins • Twin management require multidisciplinary approach
Editor's Notes
- #27: nausea and vomiting occurs with increased frequency and severity Prevalence of anaemia is more due to greater blood volume expansion of 50-60%, red cell mass and increase iron and folate requirements Unexplained weight gain not explained by pe or obesity Bp- early in pregnancy dbp will be lower than singleton pregnancy and it rises to a greater degree at term Also preeclampsia is a common association Women with twin pregnancies have levels of antiangiogenic soluble fms like tk-1(sflt-1) twice that of singleton pregnancy and decreased levels of pigf. Hypertension not only develops often but also earlier and more severe. hydramnios-mc in mz twins and usually involves 2nd sac( increased renal perfusion and urine output) Antepartum haemmorhage – increase incidence of placenta previa due to bigger size of placenta encroaching to lower segment , increased incidence of preeclampsia, sudden escape of liquor following rupture of amniotic sac
- #28: Malpresentation –more common in second baby Preterm labour- due to overdistension of uterus hydramnios and prom Mechanical distress , palpitation, dyspnea , varicosities and haemmorhoids may be increased as compared to singleton pregnancy gdm
- #29: Early rupture of membrane and cord proplapse –increased in second baby Prolonged labour Increased operative interference due to malpresentation Intrapartum bleeding following birth of first baby due to separation of placenta following reduction of placental site Pph due to atony of uterus because of overdistension , longer time taken by big placenta to separate, bigger surface area of placenta exposing more sinuses , implantation of placenta in lower segment which is less retractile
- #31: Size discordancy in dizygotic usually results from unequal placentation with one placental site receiving more perfusion than others. Degree of growth restriction in monozygotic twins is greater ..generally due to unequal allocation of blastomeres, unequal distribution of nutrients and oxygen within a single placenta and discordant structural anomalies.

















































![Discordant Twin
• Significant intrauterine fetal size discordance in monochorionic twins:
Difference in estimated fetal weight [EFW] of greater than 20%
and
Smaller twin with EFW or abdominal circumference (AC) on ultrasound
of < 10th centile for gestation
• It is associated with marginally increased perinatal risk
• Calculate percentage EFW discordance using the following formula:
[larger twin EFW—smaller twin EFW]/larger twin EFW) × 100](https://image.slidesharecdn.com/monochorionictwinpregnancy1-250826144107-1ed90754/85/MONOCHORIONIC-TWIN-PREGNANCY-detailed-pptx-52-320.jpg)

![Diagnosis of Selective Growth Restriction (sGR)
Estimated fetal weight [EFW] of one twin is less than 3rd centile
OR
there are 2 of the following 3 parameters—
(i) A growth discordance of greater than 25% difference in EFW between
the fetuses
(ii) The smaller fetus having an EFW or AC less than 10th centile for
gestation
(iii) Abnormal umbilical artery (UA) Doppler of the smaller fetus (UA-PI
of greater than 95th centile or shows absent or reversed end-diastolic
velocity)](https://image.slidesharecdn.com/monochorionictwinpregnancy1-250826144107-1ed90754/85/MONOCHORIONIC-TWIN-PREGNANCY-detailed-pptx-54-320.jpg)
























![TAPS Management
• Care options may include:
(i) Conservative management with close observation and surveillance
(ii) In utero transfusion [of the anaemic twin] with or without exchange
transfusion [of the polycythaemic fetus]
(iii) Fetoscopic laser ablation
(iv) Selective termination of pregnancy (in some circumstances)](https://image.slidesharecdn.com/monochorionictwinpregnancy1-250826144107-1ed90754/85/MONOCHORIONIC-TWIN-PREGNANCY-detailed-pptx-80-320.jpg)













